 MGMT Orthopedic Implants
MGMT Orthopedic Implants
 MGMT Orthopedic Implants
MGMT Orthopedic Implants
 Surgery Orthopedic Implants Cannulated Locking Compression Screw
Surgery Orthopedic Implants Cannulated Locking Compression Screw
 Medical OEM&ODM Titanium Cannulated Headless Compression Screw
Medical OEM&ODM Titanium Cannulated Headless Compression Screw
 Headless Compression Hollow Screw Cannulated Bone Screws
Headless Compression Hollow Screw Cannulated Bone Screws
 Bone Cement-Injectable Cannulated Pedicle Screws
Bone Cement-Injectable Cannulated Pedicle Screws
 Orthopedic Cannulated Screw for Bone Fixation Trauma Implant
Orthopedic Cannulated Screw for Bone Fixation Trauma Implant
 Orthopedic Surgical ACL Surgery Arthroscopy Instrument Set
Orthopedic Surgical ACL Surgery Arthroscopy Instrument Set
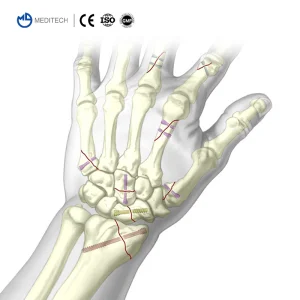 Titanium Medical Bone Screws Herbert Cannulated Compression Screw
Titanium Medical Bone Screws Herbert Cannulated Compression Screw
 Nx Medical Cannulated Conical Headless Compression Screw
Nx Medical Cannulated Conical Headless Compression Screw
The global medical device market is currently witnessing a transformative phase, particularly in the realm of Orthopedic Cannulated Drills and their associated implant systems. As surgical procedures move toward Minimally Invasive Surgery (MIS), the demand for precision-engineered cannulated tools has skyrocketed. These instruments are no longer just simple drills; they are sophisticated components of a larger surgical ecosystem that allows surgeons to perform complex bone fixations through tiny incisions, guided by wires.
From the trauma centers of North America to the emerging private hospitals in Southeast Asia, the commercial demand for high-durability, biocompatible cannulated instruments is at an all-time high. China, as a global manufacturing powerhouse, has evolved from a simple assembly point to a center of high-end innovation, rivaling traditional Western manufacturers in both quality and technological integration.
Integrated supply chains in China allow for rapid prototyping and large-scale manufacturing of medical-grade titanium and stainless steel components.
Leading manufacturers now strictly adhere to ISO 13485 and CE certifications, ensuring safety across all international borders.
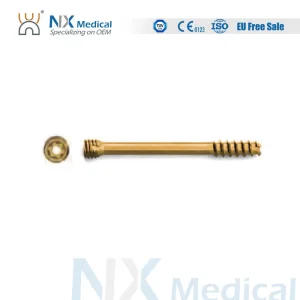
Our flagship Nx Medical Orthopedic Implant Cannulated Headless Compression Screw represents the pinnacle of hollow-core technology. Designed for small bone fragments and intra-articular fractures, this product demonstrates the high level of machining precision our factory achieves.
By utilizing advanced CNC 5-axis machining, we ensure that the cannulation is perfectly centered, allowing for seamless guidewire insertion and reducing surgical time in critical trauma scenarios. This is a core reason why global distributors prefer our Custom Cannulated Screw Solutions.
The future of orthopedic cannulated drills lies in their compatibility with robotic surgical assistants. Our R&D department is currently focusing on "Smart Drills" that provide haptic feedback and precise depth control to prevent cortical breakthrough.
While Titanium remains king, there is a growing trend toward magnesium-based biodegradable cannulated screws that disappear once the bone has healed, eliminating the need for a second surgery to remove implants.
Patient-specific cannulated guides and drills are becoming standard. We leverage AI-driven design tools to create instruments that match the unique anatomy of patients, improving successful outcome rates in complex spinal surgeries.
In high-volume trauma centers, reliability is paramount. Our cannulated drills are designed for heavy-duty sterilization cycles without compromising the sharpness of the cutting edge or the integrity of the motor housing.
Portability and battery life are the key procurement drivers for defense medical agencies. We provide lightweight, high-torque cannulated power tools that perform reliably in challenging environments.
Global procurement officers are increasingly looking for Contract Manufacturing Partners rather than just vendors. This shift is driven by the need for supply chain resilience. By partnering with a leading China manufacturer like MGMT, businesses gain access to a vertically integrated facility that handles everything from raw titanium sourcing to final sterile packaging.
Whether it's a 6.5mm Cannulated Screw Instrument Set for a large orthopedic hospital or a custom OEM project for a medical startup, our ability to localize production requirements to meet specific national standards (such as FDA or NMPA) makes us the ideal exporter for the modern age.
Efficiency in the operating room is determined by the quality of the tools. Our 6.5mm Cannulated Screw Instrument Set is a comprehensive solution for orthopedic surgeons. It includes everything from the initial guide wire to the final compression screw driver, all organized in a high-grade sterilization box.
This set is engineered to withstand the rigorous demands of modern orthopedics, ensuring that every drill bit and tap maintains its precision over hundreds of uses. Explore our Surgical Set Specifications for more details.

As a leading OEM/ODM manufacturer specializing in orthopedic instruments, we are your one-stop shop for comprehensive medical device contract manufacturing solutions. Our expertise lies in crafting highly efficient, innovative, and fully customized orthopedic medical devices. By leveraging advanced technologies and a deep understanding of industry needs, our divisions will partner with you to refine your designs, improving performance and reducing cost while ensuring compliance with established and emerging regulations. Trust us to transform your concepts into state-of-the-art orthopedic products that enhance patient care and drive your business forward.
Precision Medical Manufacturing: MGMT invests strategically in state-of-the-art manufacturing technologies to deliver unmatched precision machining capabilities for orthopedic medical instruments. Our commitment to innovation spans advanced CNC systems, five-axis machining centers, and automated inspection tools, enabling us to achieve micron-level tolerances critical for medical device accuracy.

Our vertically integrated approach—from design validation to rapid prototyping—accelerates time-to-market while adhering to ISO 13485 standards. Whether machining multi-faceted components or micro-machined features requiring high precision, we leverage automated quality control systems (e.g., CMM inspection) to ensure every part meets rigorous medical device requirements.




 Nx Medical Cannulated Headless Compression Screw
Nx Medical Cannulated Headless Compression Screw
 Orthopedic Implant Cannulated Headless Compression Screw - II
Orthopedic Implant Cannulated Headless Compression Screw - II
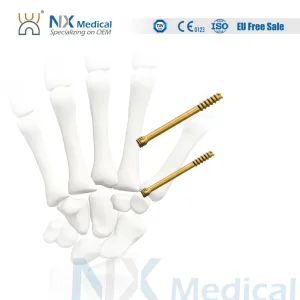 Orthopedic Implant Cannulated Headless Compression Screw
Orthopedic Implant Cannulated Headless Compression Screw
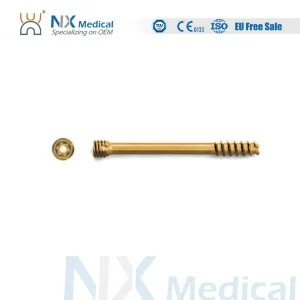 Nx Medical Cannulated Headless Compression Screw
Nx Medical Cannulated Headless Compression Screw
 6.5mm Cannulated Screw Instrument Set Orthopedic
6.5mm Cannulated Screw Instrument Set Orthopedic
 Titanium Cannulated Beleved Bunionectomy Screw
Titanium Cannulated Beleved Bunionectomy Screw
 Canwell 4.5mm/7.3mm Cannulated Screw Instrument Set
Canwell 4.5mm/7.3mm Cannulated Screw Instrument Set
 Titanium Orthopedic Implants Fixation Surgical Cannulated Screw
Titanium Orthopedic Implants Fixation Surgical Cannulated Screw