 MGMT Orthopedic Implants
MGMT Orthopedic Implants
 MGMT Orthopedic Implants
MGMT Orthopedic Implants
In the contemporary orthopedic market, Bipolar Femoral Heads represent a critical solution for femoral neck fractures, particularly in elderly patients. Unlike monopolar designs, bipolar heads feature a dual-bearing mechanism that significantly reduces acetabular wear. The global market is currently driven by an aging demographic and the rising incidence of osteoporosis-related fractures. Suppliers from Germany, the USA, and China are competing to offer the best balance between biocompatibility and biomechanical longevity.
The transition from traditional stainless steel to Cobalt-Chromium (CoCrMo) and ceramic-on-polyethylene interfaces has redefined durability. Key trends include:

Procurement teams in hospitals and distribution agencies now prioritize ISO 13485 compliance and CE/FDA certifications above all else. In localized scenarios:
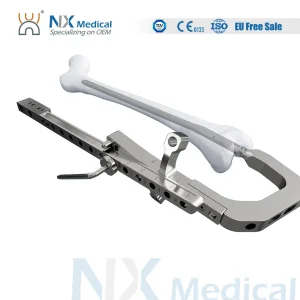
As a leading OEM/ODM manufacturer specializing in orthopedic instruments, we are your one-stop shop for comprehensive medical device contract manufacturing solutions. Our expertise lies in crafting highly efficient, innovative, and fully customized orthopedic medical devices.
MGMT invests strategically in state-of-the-art manufacturing technologies to deliver unmatched precision machining capabilities for orthopedic medical instruments. Our commitment to innovation spans advanced CNC systems, five-axis machining centers, and automated inspection tools, enabling us to achieve micron-level tolerances critical for medical device accuracy.

While the market is diverse, the top exporters generally fall into three categories:
Based in the USA and Europe, these suppliers lead in R&D and premium materials like PEEK and specialized ceramics.
Companies like MGMT provide high-precision instruments and implants with incredible efficiency and ISO-certified quality.
Focusing on emergency surgery kits, these exporters ensure that hospitals have access to a full range of sizes for acute femoral fractures.
Vertical Integration: Our approach—from design validation to rapid prototyping—accelerates time-to-market while adhering to ISO 13485 standards. Whether machining multi-faceted components or micro-machined features requiring high precision, we leverage automated quality control systems (e.g., CMM inspection) to ensure every part meets rigorous medical device requirements.