 MGMT Orthopedic Implants
MGMT Orthopedic Implants
 MGMT Orthopedic Implants
MGMT Orthopedic Implants
Advanced external fixation systems designed for small bone trauma and reconstruction.
The global market for Mini External Fixators specifically designed for hand and foot surgeries is experiencing a significant paradigm shift. As orthopedic interventions move toward minimally invasive techniques, the demand for precision-engineered mini fixators has skyrocketed. Manufacturers and exporters in this sector are no longer just component suppliers; they are critical partners in the surgical outcome chain.
In developed regions like North America and Europe, the focus is on biocompatible materials such as Titanium Grade 5 and carbon fiber composites to reduce weight while maintaining rigid stability. Meanwhile, in emerging markets across Asia-Pacific and Latin America, the primary driver is the balance between cost-efficiency and clinical reliability. As a leading China-based factory, we bridge this gap by offering high-tech manufacturing at competitive industrial scales.
Emphasis on Dynamic Fixation allowing early joint mobilization, reducing the risk of post-operative stiffness in delicate hand and foot structures.
Integration of 3D-guided placement systems and low-profile designs that minimize soft tissue irritation during long-term fixation.
Rising incidence of trauma from industrial activities and the increasing prevalence of corrective surgeries for diabetic foot complications.
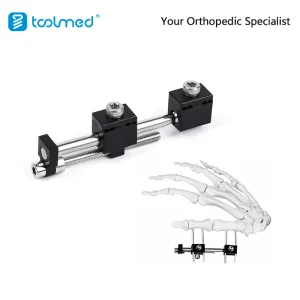
Our Small Bone Mini External Fixation System is engineered for complex orthopedic trauma. Featuring a modular design that allows for multi-planar adjustment, it provides surgeons with the flexibility needed for intricate metacarpal and phalangeal fractures.
Key Benefits:
Historically, external fixation was reserved for massive trauma. Today, the "Mini" revolution has brought these benefits to the digits. The trend is moving toward Hybrid Systems—combining internal screws with external rails to manage fragmented fractures that were once considered inoperable.
Distributors and hospitals globally are looking for "Regulatory Compliance" and "Supply Chain Resilience." With the tightening of MDR (Medical Device Regulation) in Europe and FDA requirements in the US, our role as an ISO 13485 certified manufacturer is more vital than ever. Procurement officers are prioritizing manufacturers who can provide full traceability and consistent metallurgical quality.
In urban trauma centers, mini fixators are essential for high-velocity injuries (machinery accidents). In contrast, in geriatric care, they are used for stabilizing osteoporotic bone where internal plating might fail. Our systems are also widely adopted in Veterinary Orthopedics, providing stabilization for avian and small mammal bone repairs.
China has transformed from a low-cost hub to a high-precision manufacturing powerhouse. Our factory utilizes 5-axis CNC machining and robotic polishing to ensure every Mini External Fixator meets international quality benchmarks while offering a 30-40% cost advantage over Western counterparts. This efficiency allows us to reinvest in R&D, creating patented designs for better patient outcomes.

Designed for maximum stability with minimal footprint, the Phalanges and Metacarpals Mini Fixator is the choice for surgeons requiring rigid fixation in tight anatomical spaces. Its unilateral rail system allows for compression and distraction, essential for non-union bone healing.
Precision Engineered in our ISO 13485 Facility. This product exemplifies our commitment to high-tolerance medical machining and innovative orthopedic design.
As a leading OEM/ODM manufacturer specializing in orthopedic instruments, we are your one-stop shop for comprehensive medical device contract manufacturing solutions. Our expertise lies in crafting highly efficient, innovative, and fully customized orthopedic medical devices.

MGMT invests strategically in state-of-the-art manufacturing technologies to deliver unmatched precision machining capabilities. Our commitment to innovation spans advanced CNC systems, five-axis machining centers, and automated inspection tools, enabling us to achieve micron-level tolerances critical for medical device accuracy.
Our vertically integrated approach—from design validation to rapid prototyping—accelerates time-to-market while adhering to ISO 13485 standards. Whether machining multi-faceted components or micro-machined features requiring high precision, we leverage automated quality control systems (e.g., CMM inspection) to ensure every part meets rigorous medical device requirements.




Comprehensive range of mini external fixation systems for every clinical scenario.