 MGMT Orthopedic Implants
MGMT Orthopedic Implants
 MGMT Orthopedic Implants
MGMT Orthopedic Implants
The global suture anchors market is currently undergoing a transformative phase, driven by the exponential growth of the Sports Medicine sector. As populations age and active lifestyles become more prevalent, the demand for minimally invasive surgical procedures—particularly for rotator cuff repairs, ACL reconstruction, and labral repairs—has surged. Suture anchors have become the "gold standard" for reattaching soft tissue to bone, providing the mechanical stability required for biological healing.
Market data indicates a steady CAGR of over 5.8%, with North America and Europe leading in terms of surgical volume. However, the Asia-Pacific region is rapidly emerging as a manufacturing and consumption powerhouse. As a leading Suture Anchors Supplier & Exporter, MGMT bridges the gap between high-end clinical requirements and efficient large-scale production, ensuring that medical facilities worldwide have access to premium fixation devices.
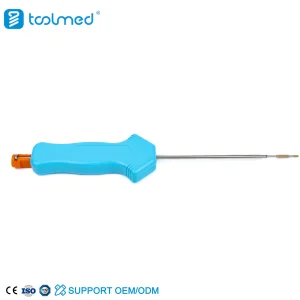
MGMT stays at the forefront of these trends by investing in R&D for Knot-Free Suture Systems and high-performance PEEK materials, ensuring our partners receive the most advanced surgical tools available.
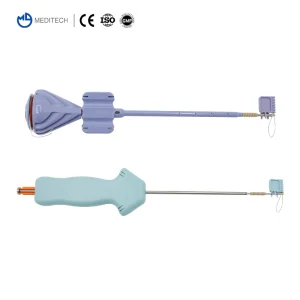
International procurement managers at major hospitals and medical device distributors prioritize three factors: Biocompatibility, Pull-out Strength, and Supply Chain Stability. Our suture anchors are engineered to meet these rigorous demands. Whether it's a specialized orthopedic clinic in Germany or a high-volume general hospital in Brazil, the need for reliable fixation remains universal.
Optimized for rotator cuff and SLAP lesions, providing high-torque stability in the humerus.
Designed for ACL/PCL reconstruction and labral refixation, focusing on long-term biological integration.
Micro-anchors designed for delicate ligament repairs, ensuring minimal bone removal.
MGMT is a leading OEM/ODM manufacturer specializing in orthopedic instruments. As your one-stop shop for comprehensive medical device contract manufacturing solutions, our expertise lies in crafting highly efficient, innovative, and fully customized orthopedic medical devices. By leveraging advanced technologies and a deep understanding of industry needs, our divisions will partner with you to refine your designs, improving performance and reducing cost while ensuring compliance with established and emerging regulations. Trust us to transform your concepts into state-of-the-art orthopedic products that enhance patient care and drive your business forward.

MGMT invests strategically in state-of-the-art manufacturing technologies to deliver unmatched precision machining capabilities for orthopedic medical instruments. Our commitment to innovation spans advanced CNC systems, five-axis machining centers, and automated inspection tools, enabling us to achieve micron-level tolerances critical for medical device accuracy.
Our vertically integrated approach—from design validation to rapid prototyping—accelerates time-to-market while adhering to ISO 13485 standards. Whether machining multi-faceted components or micro-machined features requiring high precision, we leverage automated quality control systems (e.g., CMM inspection) to ensure every part meets rigorous medical device requirements.

FT2 Workshop Area

Years Establishment

Engineer Team

Business Coverage Countries
Sourcing from a Chinese factory like MGMT offers a unique blend of cost-efficiency, manufacturing scale, and rapid innovation. Unlike traditional Western manufacturers, we provide a more agile supply chain that can pivot quickly to custom design requirements (OEM/ODM).
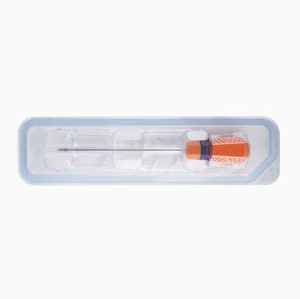
Our suture anchors are not just fasteners; they are highly engineered medical systems. The thread design is optimized for specific bone densities (cancellous vs. cortical), and the suture material (typically UHMWPE) provides the highest possible tensile strength to prevent breakage during the critical healing phase.
PEEK (Polyetheretherketone) Advantages:
Titanium Alloy (Ti6Al4V ELI) Advantages:
As a premier Suture Anchors Supplier & Exporter, our mission is to empower surgeons with tools that provide absolute confidence in fixation. From the initial 3D design phase to final sterile packaging, every step is monitored by our team of 50+ expert engineers. Our footprint spans 30+ countries, making us a trusted partner for hospitals, clinics, and medical device brands looking for excellence in sports medicine. Contact us today to learn more about our OEM/ODM capabilities and how we can support your orthopedic product lineup.