 MGMT Orthopedic Implants
MGMT Orthopedic Implants
 MGMT Orthopedic Implants
MGMT Orthopedic Implants
 Orthopedic Medical Endoscope Instruments Set Surgical in Bone Surgery Hospital OEM
Orthopedic Medical Endoscope Instruments Set Surgical in Bone Surgery Hospital OEM Surgical Instrument Set Medical Equipment Orthopedic Acl Instrument
Surgical Instrument Set Medical Equipment Orthopedic Acl Instrument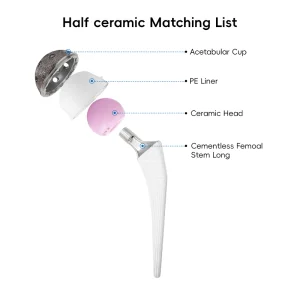 Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement
Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement Replacement CE Sterilized, Carton Package Prosthesis Knee Joint
Replacement CE Sterilized, Carton Package Prosthesis Knee Joint CE Certified Orthopedic Expert Femoral Instrument Set for Shoulder Surgery Essential Orthopedic Surgical Instruments
CE Certified Orthopedic Expert Femoral Instrument Set for Shoulder Surgery Essential Orthopedic Surgical Instruments Orthopedic Toolmed Knee Prosthesis Manufacturer Knee Replacement Ortho Surgery Arthroplasty Tka
Orthopedic Toolmed Knee Prosthesis Manufacturer Knee Replacement Ortho Surgery Arthroplasty Tka Jiangsu Replacement Sterilized Package Prosthesis Knee Joint
Jiangsu Replacement Sterilized Package Prosthesis Knee Joint S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty SurgeryThe global orthopedic market is witnessing a significant surge in demand for Revision Hip/Knee Systems. As primary joint replacements performed in the early 2000s reach their end-of-life cycle, and with an increasingly active aging population, the clinical necessity for revision surgery has moved to the forefront of orthopedic care. Currently, revision procedures account for approximately 10-15% of all joint replacements globally, creating a multi-billion dollar industrial sector focused on biocompatibility, modularity, and long-term fixation.
Major markets in North America and Europe are leading the procurement trends, focusing on bone-stock preservation and infection management. Meanwhile, emerging markets in Asia and Latin America are rapidly adopting these advanced systems to improve post-operative outcomes in secondary surgeries. As a dedicated Revision Hip/Knee Systems Supplier & Exporter, we bridge the gap between high-precision engineering and clinical availability.
As a leading OEM/ODM manufacturer specializing in orthopedic instruments,we are your one - stop shop for comprehensive medical device contract manufacturing solutions. Our expertise lies in crafting highly efficient, innovative, and fully customized orthopedic medical devices. By leveraging advanced technologies and a deep understanding of industry needs, our divisions will partner with you to refine your designs, improving performance and reducing cost while ensuring compliance with established and emerging regulations. Trust us to transform your concepts into state-of-the-art orthopedic products that enhance patient care and drive your business forward.

Modern revision systems utilize modular stems and augments to address massive bone loss, allowing surgeons to customize the implant to the patient's unique anatomy intraoperatively.
Additive manufacturing is revolutionizing cementless fixation. Porous structures mimic human trabecular bone, encouraging biological ingrowth for superior stability.
The integration of digital navigation and robotic systems ensures precise component positioning, which is critical in revision cases where landmarks are often compromised.
Global hospital chains and medical distributors are increasingly seeking Revision Hip/Knee Systems that offer a balance between clinical performance and economic viability. Procurement departments now prioritize suppliers who provide comprehensive instrument sets along with the implants to minimize surgical delays.
In local application scenarios, such as specialized trauma centers or university hospitals, the ability to provide "one-stop" solutions—including pulsed lavage systems and specialized extraction tools—is a significant competitive advantage. Our role as a global exporter ensures that even the most remote surgical centers have access to CE-certified, high-grade orthopedic technology.
China has evolved into a global epicenter for high-end medical device manufacturing. Our facility leverages the country's robust industrial supply chain and advanced metallurgical expertise. As a leading Revision Hip/Knee Systems Supplier, we offer:
1. Unmatched Precision: Utilizing 5-axis CNC machining centers from Germany and Japan, we achieve tolerances within microns, essential for the fit and finish of revision components.
2. Cost Efficiency: Through scale and vertically integrated production—from raw material sourcing to final sterilization—we provide high-quality implants at a fraction of the cost of Western competitors.
3. Rapid Prototyping: Our R&D team can transform clinical feedback into modified designs within weeks, not months, ensuring our products stay at the cutting edge of surgical trends.
Furthermore, our strict adherence to ISO 13485 and international regulatory standards ensures that every product exported meets the rigorous safety requirements of the global medical market. From cementless titanium stems to specialized surgical instrumentation, our "Made in China" mark stands for precision and reliability.
MGMT invests strategically in state-of-the-art manufacturing technologies to deliver unmatched precision machining capabilities for orthopedic medical instruments. Our commitment to innovation spans advanced CNC systems, five-axis machining centers, and automated inspection tools, enabling us to achieve micron-level tolerances critical for medical device accuracy. Our vertically integrated approach—from design validation to rapid prototyping—accelerates time-to-market while adhering to ISO 13485 standards. Whether machining multi-faceted components or micro-machined features requiring high precision, we leverage automated quality control systems (e.g., CMM inspection) to ensure every part meets rigorous medical device requirements.
The complexity of revision arthroplasty requires a systematic approach to implant selection. Our Revision Hip Systems are designed with a focus on distal fixation, utilizing tapered fluted stems that provide rotational stability even in the presence of proximal femoral defects. The use of high-strength Titanium-6Aluminum-4Vanadium alloy ensures fatigue resistance while maintaining an elastic modulus closer to that of cortical bone compared to Cobalt-Chrome alloys.
In the realm of Revision Knee Systems, addressing ligamentous instability is paramount. Our constrained and semi-constrained liners are engineered from Ultra-High-Molecular-Weight Polyethylene (UHMWPE), often infused with Vitamin E to resist oxidative wear. This material science innovation is a cornerstone of our export strategy, offering long-term solutions for patients worldwide. As a supplier, we provide not just the implants, but the entire surgical ecosystem, including trial components and precision instrumentation that facilitate the "femur-first" or "tibia-first" surgical techniques favoured by modern surgeons.
 Disposable Pulsed Lavage System
Disposable Pulsed Lavage System Orthopedic Equipment for Bone Surgery Cage System Medical Implement
Orthopedic Equipment for Bone Surgery Cage System Medical Implement Orthopedic Mini Instrument for Hand and Foot Surgery Instrument Set Surgical
Orthopedic Mini Instrument for Hand and Foot Surgery Instrument Set Surgical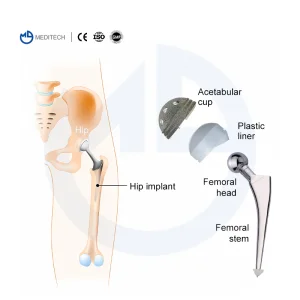 Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement Orthopedic Surgical Instrument Set for Orthopaedic Cable Instrument for Bone Surgery
Orthopedic Surgical Instrument Set for Orthopaedic Cable Instrument for Bone Surgery Proximal Lateral Femoral Plate -Orthopedic Implant System for Femoral Shaft or Condylar Fracture Surgery
Proximal Lateral Femoral Plate -Orthopedic Implant System for Femoral Shaft or Condylar Fracture Surgery CE Prosthesis Replacement Sterilized, Carton Package Class III Knee Joint
CE Prosthesis Replacement Sterilized, Carton Package Class III Knee Joint OEM Orthopedic Surgical Instrument Femoral Condyle Replacement Artificial Knee Joint Implants
OEM Orthopedic Surgical Instrument Femoral Condyle Replacement Artificial Knee Joint Implants