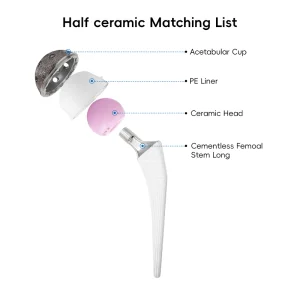
Expert Insights: Choosing the Right Cementless Hip Stem Factory
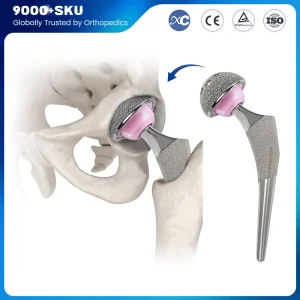
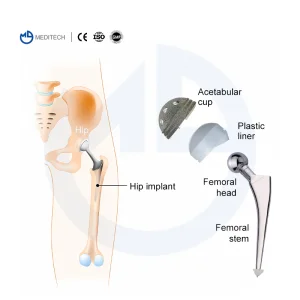
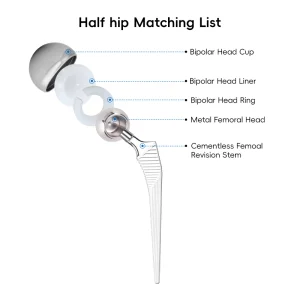
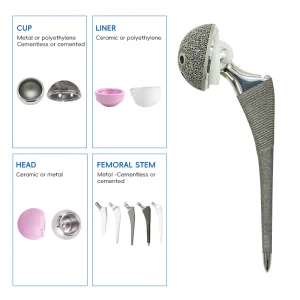
When sourcing Cementless Hip Stems from China, technical specifications are paramount. A high-quality stem must exhibit excellent biocompatibility, mechanical strength, and surface roughness. Our manufacturing process begins with medical-grade Titanium alloy (Ti6Al4V), known for its high strength-to-weight ratio and corrosion resistance. Using advanced forging or casting methods, we create the blank which is then precision-machined.
The "Cementless" aspect is achieved through sophisticated surface treatments. Our factory utilizes Vacuum Plasma Spraying (VPS) to apply a porous titanium layer. This layer creates a 3D environment that encourages bone cells to migrate and anchor directly into the implant, creating a permanent biological bond. This is often supplemented with a Hydroxyapatite (HA) coating, a natural bone mineral that further stimulates osteoblast activity.
Manufacturing Precision & Quality Control
At MGMT, we understand that an implant is only as good as its fit. Our 5-axis CNC machining centers allow us to manufacture complex geometries, including tapered designs that ensure a perfect "wedge fit" in the femoral canal. Each batch of Cementless Hip Stems undergoes rigorous quality checks:
- Dimensional verification using CMM (Coordinate Measuring Machines).
- Surface roughness testing to ensure optimal coating adhesion.
- Fatigue testing to simulate decades of physiological loading.
- Sterilization validation to ensure patient safety.
Furthermore, our vertically integrated approach—from design validation to rapid prototyping—accelerates time-to-market while adhering to ISO 13485 standards. Whether machining multi-faceted components or micro-machined features requiring high precision, we leverage automated quality control systems to ensure every part meets rigorous medical device requirements.
The Future of Orthopedic Partnerships
In the coming years, the trend toward personalized medicine will drive the demand for patient-specific implants. As a forward-thinking China Hip Stem factory, we are investing in 3D printing (Additive Manufacturing) technologies to produce custom stems for complex anatomical cases. This commitment to innovation makes us more than just a supplier; we are a strategic partner in the global orthopedic industry.
By choosing a professional manufacturer like MGMT, you gain access to a world-class engineering team, state-of-the-art facilities, and a cost structure that allows you to remain competitive in a challenging healthcare market. We invite global partners to explore our facility and witness the precision that goes into every single implant we produce.
 MGMT Orthopedic Implants
MGMT Orthopedic Implants