Strategic Supply Chain for Orthopedic Implants in France
Navigating the medical device market in France requires more than just high-quality products; it requires a deep understanding of the regulatory landscape and the logistical demands of the Paris healthcare network. As a leading Intramedullary Nails Factory, we provide a streamlined supply chain that bridges the gap between precision manufacturing and surgical application.
Why Paris-Based Distributors Choose Our Implants
Distributors in the Paris region look for reliability, sterile packaging options, and a comprehensive range of sizes. Our factory supports these needs through high-volume production capabilities without compromising on the custom requirements of the French market. We offer:
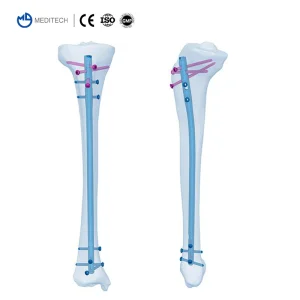
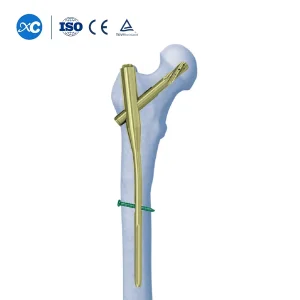
- Comprehensive Titanium Interlocking Nail systems for Femur, Tibia, and Humerus.
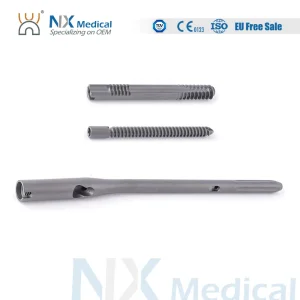
- Surgical instrument sets designed for ergonomic use in high-pressure Parisian operating rooms.
- OEM services for French medical brands looking to expand their portfolio with CE-marked implants.
The Clinical Importance of Design in Modern Orthopedics
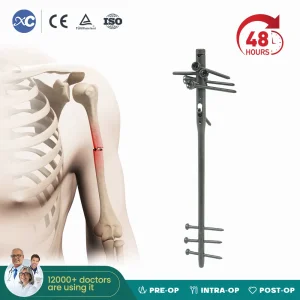
Intramedullary nailing has become the gold standard for many long-bone fractures. In Paris, the emphasis is on biological fixation. By utilizing an intramedullary nail, surgeons can achieve stable fixation while preserving the periosteal blood supply, which is crucial for fracture healing. Our nails, such as the X Series Femoral Nail, feature anatomical curvatures and locking options that cater to the diverse morphology of the European patient population.
Furthermore, the color-coded systems we provide (Blue, Green, Gold) are not merely aesthetic. They serve as a critical safety feature in the surgical suite, allowing for rapid identification of nail diameters and lengths, thereby reducing surgical time and anesthesia exposure for the patient—a key metric for quality of care in hospitals like Bichat–Claude Bernard.
Future-Proofing Orthopedic Care
As we look toward the future of orthopedics in Paris, the integration of 3D printing for custom jigs and the use of bio-absorbable materials are on the horizon. Our manufacturing facility is already exploring these frontiers, ensuring that our partners in France have access to the most advanced fixation technology available. We are committed to being more than just a supplier; we are a partner in the advancement of French surgical science.
 MGMT Orthopedic Implants
MGMT Orthopedic Implants