 MGMT Orthopedic Implants
MGMT Orthopedic Implants
 MGMT Orthopedic Implants
MGMT Orthopedic Implants
Toronto, Ontario, serves as a global powerhouse in medical research and surgical innovation. As a leading Intramedullary Nails Supplier & Exporter in Toronto, we operate at the intersection of Canada's most advanced healthcare corridor. The Greater Toronto Area (GTA) is home to world-renowned institutions like Sunnybrook Health Sciences Centre—Canada’s largest trauma center—and the University Health Network (UHN), which drive the demand for high-performance orthopedic implants.
The industrial landscape in Toronto for intramedullary (IM) nails is characterized by a shift toward smart manufacturing and biocompatible materials. With the rise of the MaRS Discovery District, Toronto has become a testing ground for next-generation surgical alloys. Our products are designed to meet the rigorous standards expected by Canadian orthopedic surgeons, focusing on reducing "time-in-OR" through intuitive instrumentation and superior interlocking mechanisms. As exporters, we leverage Toronto's Pearson International Airport and integrated logistics networks to ensure that our titanium and stainless steel IM nails reach clinics in Europe, Asia, and the Americas with unparalleled speed.
Utilizing Medical Grade 5 Titanium (Ti6Al4V ELI) and high-strength Stainless Steel (316LVM) to ensure maximum biocompatibility and fatigue resistance for Toronto's diverse patient demographics.
Our design process incorporates AI-assisted load-bearing simulations, ensuring each intramedullary nail can withstand the dynamic stresses of weight-bearing recovery in trauma patients.
Strategically headquartered in Toronto, we navigate complex international regulatory frameworks, providing CE and ISO-certified implants to the global market.
As a leading OEM/ODM manufacturer specializing in orthopedic instruments, we are your one-stop shop for comprehensive medical device contract manufacturing solutions. Our expertise lies in crafting highly efficient, innovative, and fully customized orthopedic medical devices. By leveraging advanced technologies and a deep understanding of industry needs, our divisions will partner with you to refine your designs, improving performance and reducing cost while ensuring compliance with established and emerging regulations. Trust us to transform your concepts into state-of-the-art orthopedic products that enhance patient care and drive your business forward.
MGMT invests strategically in state-of-the-art manufacturing technologies to deliver unmatched precision machining capabilities for orthopedic medical instruments. Our commitment to innovation spans advanced CNC systems, five-axis machining centers, and automated inspection tools, enabling us to achieve micron-level tolerances critical for medical device accuracy.

Our vertically integrated approach—from design validation to rapid prototyping—accelerates time-to-market while adhering to ISO 13485 standards. Whether machining multi-faceted components or micro-machined features requiring high precision, we leverage automated quality control systems (e.g., CMM inspection) to ensure every part meets rigorous medical device requirements.




The orthopedic industry in Toronto is currently witnessing a paradigm shift toward minimally invasive trauma surgery (MITS). Traditional open reduction techniques are being replaced by percutaneous insertion of intramedullary nails, a trend heavily supported by the teaching hospitals affiliated with the University of Toronto. Our intramedullary nails are designed to accommodate this shift, featuring radiolucent targeting handles and ultra-slim nail profiles that minimize soft tissue disruption.
Another significant trend in the Ontario market is the customization of implant colors and finishes. While aesthetic, the color-coding of nails (Blue, Green, Gold) serves a critical functional role in the operating room, allowing surgical nurses and technicians to quickly identify diameters and lengths, thereby reducing surgical errors and improving "Patient Safety Outcomes"—a key metric for Health Canada audits.
As a dedicated Intramedullary Nails Supplier in Toronto, we understand that "One Size Does Not Fit All." Our R&D team works closely with local Canadian clinicians to iterate on nail curvatures and locking hole configurations, ensuring our products are optimized for the North American skeletal morphology.
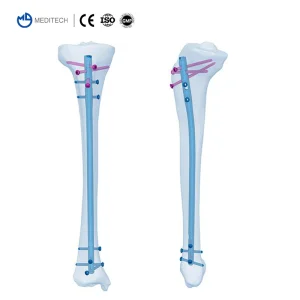
Direct Sale Orthopedic Implant Titanium Expert Tibia Interlocking Intramedullary Nail

Orthopedic Implants Reversed Femoral Intramedullary / Interlocking Nails

Surgical Instrument Professional X Series Femoral Intramedullary Nail (Reconstruction)

Best Price Intramedullary Nail System Expert Tibial Proximal Interlocking Nail

Orthopedic Implant Interlocking Nail Standard Intertan Intramedullary Nail
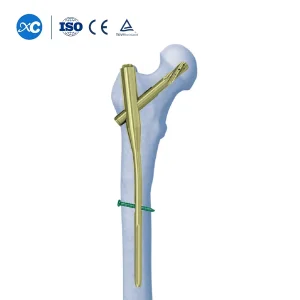
Price Gama Nails Pfna Surgery Nail Antirotation Titanium Femur Intramedullary Nail
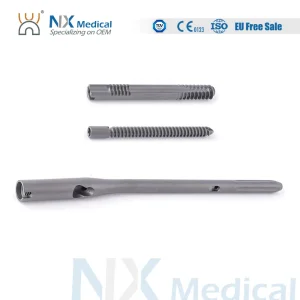
Nx Medical Pfia Nail System Titanium Intramedullary Interlocking Femoral Nail
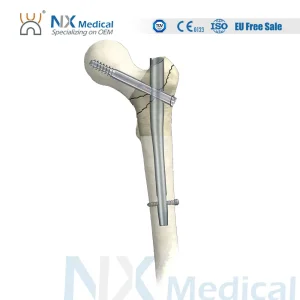
Nx Medical Orthopedic Trauma Titanium Intramedullary Interlocking Femoral Pfna Bone Nail
Managing an orthopedic supply chain requires more than just high-quality hardware; it requires a partner who understands the nuances of Regulatory Compliance. In Canada, the Medical Devices Regulations (MDR) under the Food and Drugs Act set a high bar for safety. By choosing a Toronto-focused supplier, you benefit from products that are designed with these global standards as a baseline. Our manufacturing facilities utilize high-precision CNC multi-axis milling to ensure that every interlocking hole is perfectly aligned—reducing the risk of "missed screws" during distal locking, a common frustration for surgeons.
Furthermore, our commitment to Titanium Excellence is unmatched. Titanium's modulus of elasticity is closer to human bone than stainless steel, which significantly reduces the "stress shielding" effect. This leads to better long-term bone remodeling and healthier patient outcomes. For hospitals in Toronto and across the globe, this means lower revision rates and higher patient satisfaction scores.
Our export team is fluent in the logistics of Intermodal Transport. From our warehouse in the GTA, we can coordinate air freight via Pearson (YYZ) for urgent trauma cases or sea freight for bulk inventory replenishment. Every shipment is accompanied by complete traceability documentation, including material mill certificates and sterilization records (where applicable), ensuring that your facility remains compliant with local health authority requirements.
Every intramedullary nail that leaves our facility undergoes a multi-stage inspection process. This includes Spectrographic Analysis to verify alloy composition, Surface Roughness Testing to ensure optimal osseointegration, and Fatigue Strength Testing to simulate years of human movement. We don't just supply nails; we supply the peace of mind that comes with knowing a life-saving implant is engineered to the highest possible standards.
Ready to upgrade your orthopedic inventory with Toronto's finest intramedullary solutions?
Send Inquiry NowLooking for a reliable Intramedullary Nails Supplier & Exporter in Toronto? We provide titanium intramedullary nails, interlocking nails for femoral, tibial, and humeral fractures. Our orthopedic implants are ISO 13485 certified. We serve hospitals in Toronto, Mississauga, Brampton, and export worldwide. Trauma surgery implants, medical device manufacturing Toronto, Canada orthopedic supply chain. Expert femoral nail, PFNA nail, Intertan nail, and surgical instruments contract manufacturing. Precision machining for medical devices in Ontario. High-quality surgical grade titanium implants for trauma hospitals.