 MGMT Orthopedic Implants
MGMT Orthopedic Implants
 MGMT Orthopedic Implants
MGMT Orthopedic Implants
 Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System Surgical Instrument Set
Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System Surgical Instrument Set
 Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set
Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set
 High-Strength Cocrmo Thoracolumbar Pedicle Screw and Rod System for Brazil Clinics
High-Strength Cocrmo Thoracolumbar Pedicle Screw and Rod System for Brazil Clinics
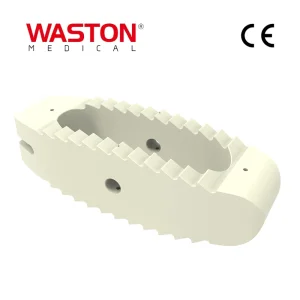 Disposable Surgical Neulen Laminoplasty Thread Inter Reduction Pedicle Screw Orthopaedic Instrument Implant O-Max Cage System
Disposable Surgical Neulen Laminoplasty Thread Inter Reduction Pedicle Screw Orthopaedic Instrument Implant O-Max Cage System
As the largest economy in South America, Brazil has established itself as a critical hub for the medical device industry, particularly in the specialized field of orthopedic spinal implants. The demand for Pedicle Screw Systems in Brazil has seen exponential growth over the last decade, driven by an aging population, an increase in trauma cases related to urbanization, and a robust private healthcare sector. For international manufacturers and local factories, Brazil represents not just a market, but a strategic production and distribution gateway for the entire MERCOSUR region.
The industrial heartbeat of the Brazilian orthopedic sector is centered in the state of São Paulo, specifically within the "Medical Valley" of Ribeirão Preto. This region hosts numerous manufacturing facilities that specialize in high-precision machining and titanium alloy processing. As a pedicle screw systems manufacturer and factory in Brazil, local entities must navigate the complex yet rewarding landscape of ANVISA (Agência Nacional de Vigilância Sanitária) regulations. These regulations ensure that every spinal fixation rod, polyaxial screw, and interbody cage meets the highest safety standards, comparable to the FDA or CE certifications.
Our factory leverages this local expertise, combining it with global innovation to provide Brazil-specific surgical solutions. By understanding the unique anatomical requirements and surgical preferences of Brazilian orthopedic surgeons, we produce pedicle screw systems that offer superior biomechanical stability and ease of implementation in diverse clinical settings, from high-end private hospitals in Rio de Janeiro to public SUS (Sistema Único de Saúde) facilities across the country.
The Brazilian orthopedic community is rapidly adopting Minimally Invasive Surgery (MIS) techniques. This shift has fundamentally changed the design requirements for pedicle screw systems. Manufacturers are now focusing on cannulated screws, specialized retractor systems, and percutaneous rod insertion tools. In Brazil, the move towards MIS is motivated by the need to reduce hospital stay durations and minimize postoperative complications, which is a priority for both private insurance providers and the public health budget.
Advanced manufacturing technologies, such as 5-axis CNC machining and 3-D titanium printing, are being integrated into Brazilian production lines. These technologies allow for the creation of complex screw thread geometries and porous surfaces that enhance osseointegration. As a forward-thinking factory, we stay ahead of these trends by investing in R&D that specifically addresses the challenges of degenerative disc disease and complex spinal deformities prevalent in the Brazilian demographic.




Operating as a pedicle screw systems factory in Brazil requires an unwavering commitment to quality. Materials used, such as Grade 5 Titanium (Ti6Al4V) and Cobalt-Chrome-Molybdenum (CoCrMo), must be sourced from certified suppliers to ensure biocompatibility and fatigue strength. In Brazil, the regulatory framework managed by ANVISA is notoriously stringent. Our manufacturing processes are ISO 13485 certified, ensuring that every production lot undergoes rigorous mechanical testing, including static and dynamic compression, tension, and torsion tests according to ASTM F1717 standards.
Furthermore, the localized application of these systems in Brazil often involves treating severe scoliosis and kyphosis. Our pedicle screw systems are designed with low-profile heads to minimize soft tissue irritation, a common concern among Brazilian surgeons. The "domino" connectors and cross-link systems we manufacture provide the additional rotational stability required for long-construct spinal surgeries.
MGMT stands as your preferred medical instruments contract manufacturing partner. As a leading OEM/ODM manufacturer specializing in orthopedic instruments, we provide a comprehensive one-stop-shop for medical device solutions tailored for the Brazilian market. Our expertise lies in crafting highly efficient, innovative, and fully customized orthopedic medical devices. By leveraging advanced technologies and a deep understanding of industry needs, our divisions partner with you to refine designs, improving performance and reducing costs while ensuring compliance with both local ANVISA and international regulations.

Our strategic investments in state-of-the-art manufacturing technologies deliver unmatched precision machining capabilities. From five-axis machining centers to automated CMM inspection tools, we achieve the micron-level tolerances critical for spinal implants. Our vertically integrated approach—from design validation to rapid prototyping—accelerates your time-to-market in the competitive Brazilian landscape.
Looking forward, the integration of digital health and robotic-assisted surgery is set to revolutionize the Brazilian orthopedic sector. Future pedicle screw systems will likely incorporate "smart" features, such as sensors for monitoring bone healing or integrated navigation markers for robotic guidance. Our factory is already exploring these frontiers, ensuring that Brazil remains at the forefront of global spinal health innovation. Whether you are a local distributor or a hospital group, partnering with a manufacturer that understands the local nuances and global trends is key to clinical success.
 Spine Surgical Pedicle Screw Titanium Surgery Orthopedic Implants Posterior Thoracolumbar Spinal Fixation System
Spine Surgical Pedicle Screw Titanium Surgery Orthopedic Implants Posterior Thoracolumbar Spinal Fixation System
 High Quality Cannulated Axial-Rotation Pedicle Screw Orthopaedic Implant Minimally Invasive System
High Quality Cannulated Axial-Rotation Pedicle Screw Orthopaedic Implant Minimally Invasive System
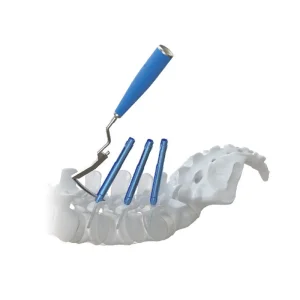 Orthopedic Pedicle Screw Spine MIS Implants System for Brazilian Hospitals
Orthopedic Pedicle Screw Spine MIS Implants System for Brazilian Hospitals
 Disposable Surgical Neulen Laminoplasty Inter Pedicle Multi Axial Screw Wave 61621 Interbody Fusion Cage System
Disposable Surgical Neulen Laminoplasty Inter Pedicle Multi Axial Screw Wave 61621 Interbody Fusion Cage System
 Disposable Titanium Spine Pedicle Screw Domino Connector Posterior Spinal Internal Fixation 5.5 System
Disposable Titanium Spine Pedicle Screw Domino Connector Posterior Spinal Internal Fixation 5.5 System
 Orthopedic Spinal Internal Fixation 6.0 System Screw Polyaxial Spine Titanium Pedicle Screws
Orthopedic Spinal Internal Fixation 6.0 System Screw Polyaxial Spine Titanium Pedicle Screws
 Quality Assurance Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Implant Arrow Cage System
Quality Assurance Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Implant Arrow Cage System
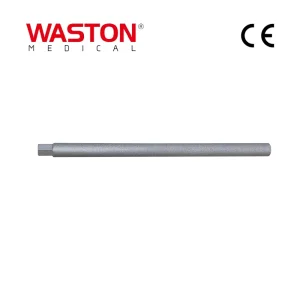 Disposable Surgical Neulen Laminoplasty Thread Inter Reduction Pedicle Screw Implant 11412 Fixation Rod IV 6.0
Disposable Surgical Neulen Laminoplasty Thread Inter Reduction Pedicle Screw Implant 11412 Fixation Rod IV 6.0