



 MGMT Orthopedic Implants
MGMT Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 400/Set Request Sample |

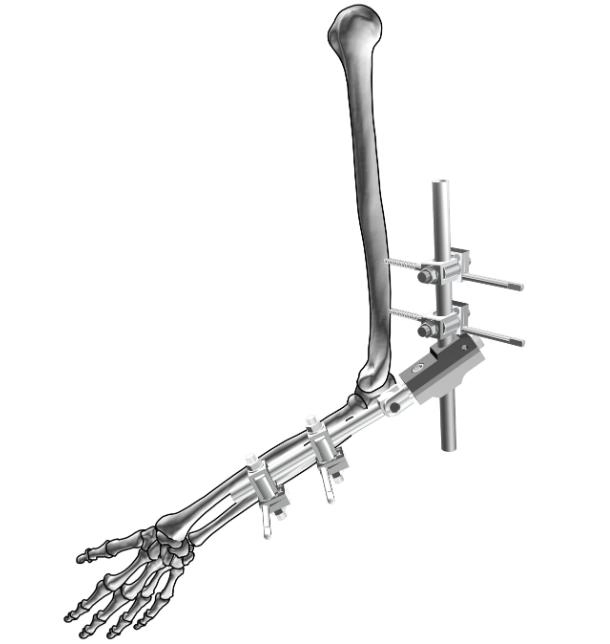
The Ilizarov External Fixator with Foot Ring is a specialized orthopedic device engineered based on the Ilizarov principle, designed for the treatment of complex foot and ankle conditions, including congenital deformities, traumatic fractures, nonunions, malunions, and post-traumatic contractures. This modular system integrates high-strength external fixation components with a customized foot ring, delivering unparalleled stability and adjustability for orthopedic surgeons worldwide.
Crafted from medical-grade stainless steel and titanium alloy, the fixator features a durable, corrosion-resistant structure. The foot ring is anatomically contoured to fit the natural curvature of the foot, providing optimal contact and support without compromising soft tissue integrity. Its modular design allows for flexible assembly, enabling surgeons to tailor the fixation configuration to each patient's unique anatomical needs.


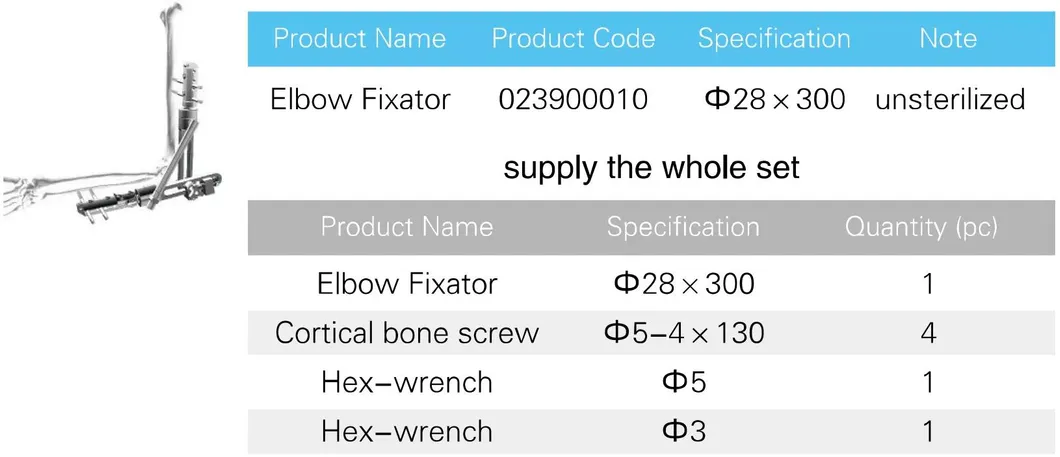
| Product Name | Specification | Size | Quantity |
|---|---|---|---|
| Dissection IV(Φ8) Femur External Fixation | Carbon Fiber Rod | Φ8×250 | 2 |
| Rod-rod Coupling | - | 4 | |
| Pin-post Coupling | - | 2 | |
| Straight Post/30°Angled Post | - | 4 | |
| One Step Pin | Φ5×180 | 4 |



