




 MGMT Orthopedic Implants
MGMT Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 100/Piece Request Sample |

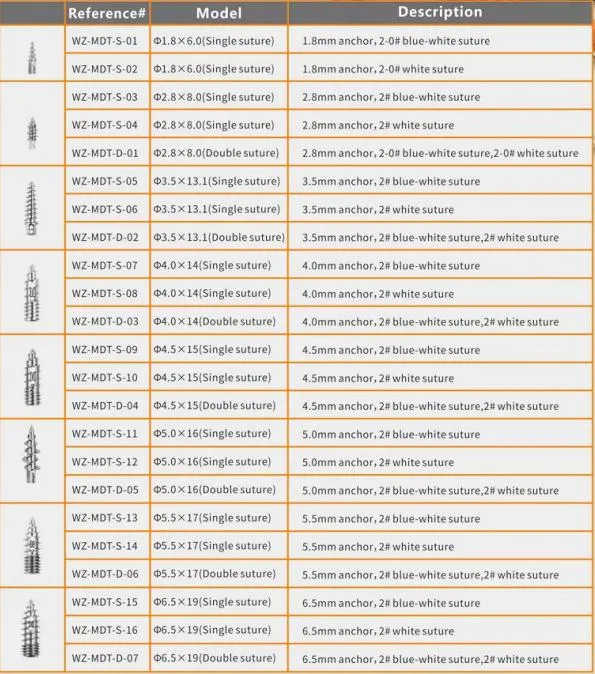
| Product Name | Non-bioabsorbable Suture Anchor |
| Ctn Size | 42*22.5*25.5CM |
| G.W./CTN | 1.5KG-1.8KG |
| Certificates | CE & ISO13485 |
| Validity | 3 Years |
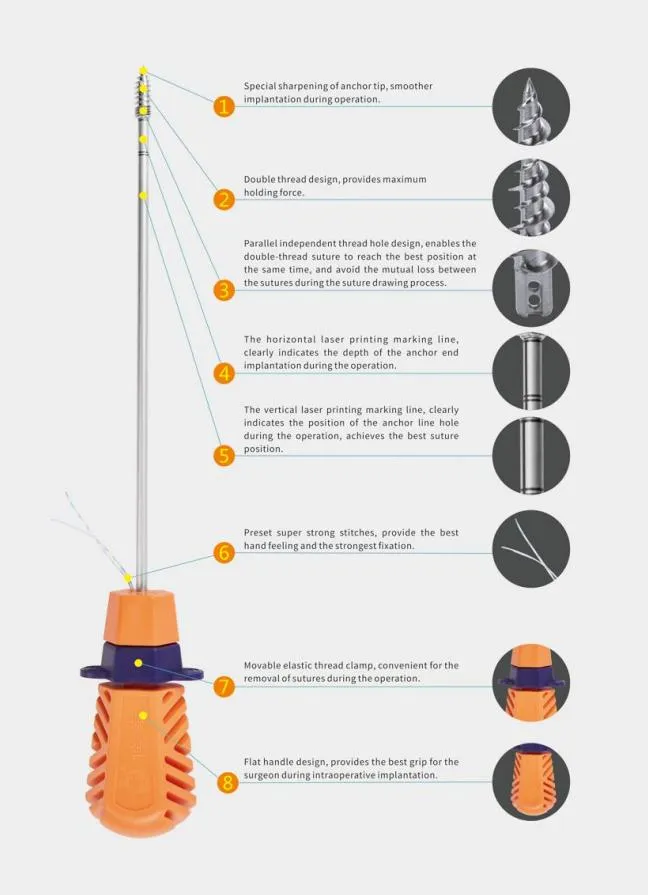
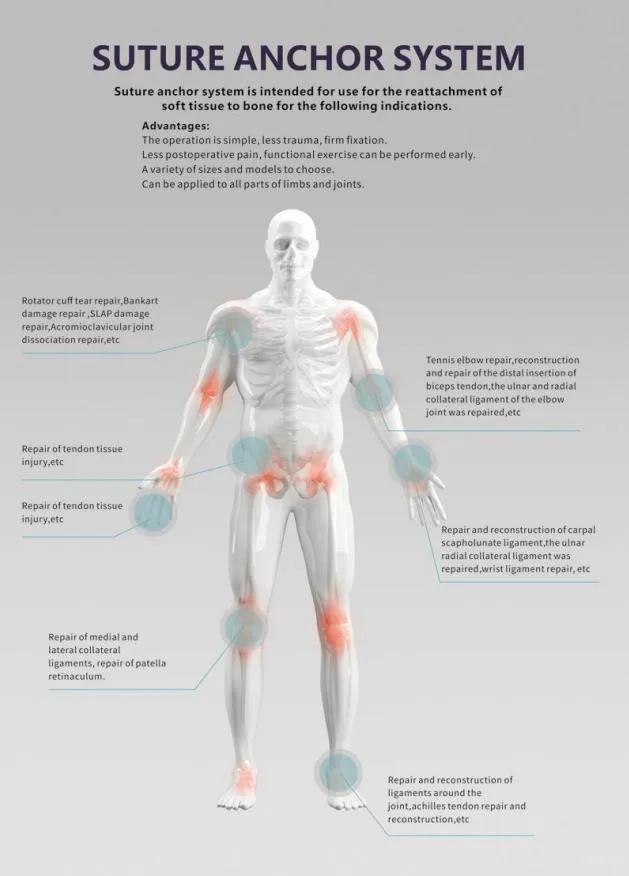



Our Surgical Lavage System is designed for effective debridement following orthopedic procedures of trauma and joint arthroplasty. All products are manufactured under strict adherence to CE and ISO13485 standards.
The system combines convenience and power for the removal of contaminated water, necrotic tissue, bone debris, wound pollutants, and foreign materials. It provides controlled pressure for efficient cleansing and irrigation on flat wound surfaces, osteotomy surfaces, or intra-medullary sites.
We continuously improve our products through clinical research and feedback from surgeons. The noise and weight (optimized to 772g) have been minimized to provide a better user experience, making it an essential medical instrument in modern orthopedic procedures.