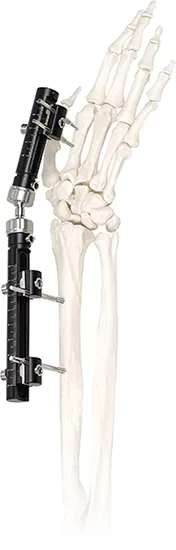
Frequently Asked Questions
What materials are used for the External Fixation system?
The system is manufactured using high-quality Titanium Alloy and Stainless Steel to ensure both durability and biocompatibility.
Is the External Fixator reusable?
Yes, the main components of the fixation system are designed to be reusable after proper sterilization procedures.
Does the system require a second surgery for removal?
No, one of the primary benefits is that it can be removed in a clinic setting without requiring a second invasive surgical procedure.
What certifications does this medical device hold?
This external fixation system is fully certified with CE and ISO13485 standards, ensuring compliance with international medical quality requirements.
How does the dynamic design assist in healing?
The controllable dynamic design allows for micro-movements consistent with the bone shaft, which helps promote bone union and faster healing.
What is the production capacity for these units?
We have a stable production capacity of 500,000 pieces per year to meet large-scale medical supply needs globally.





 MGMT Orthopedic Implants
MGMT Orthopedic Implants