

 MGMT Orthopedic Implants
MGMT Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 119/Piece Request Sample |
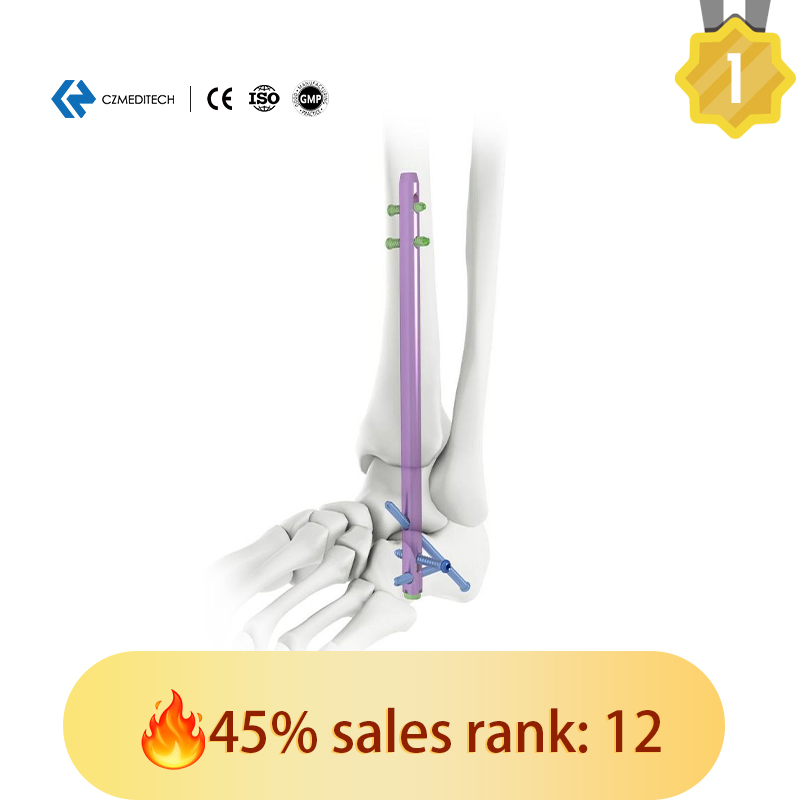
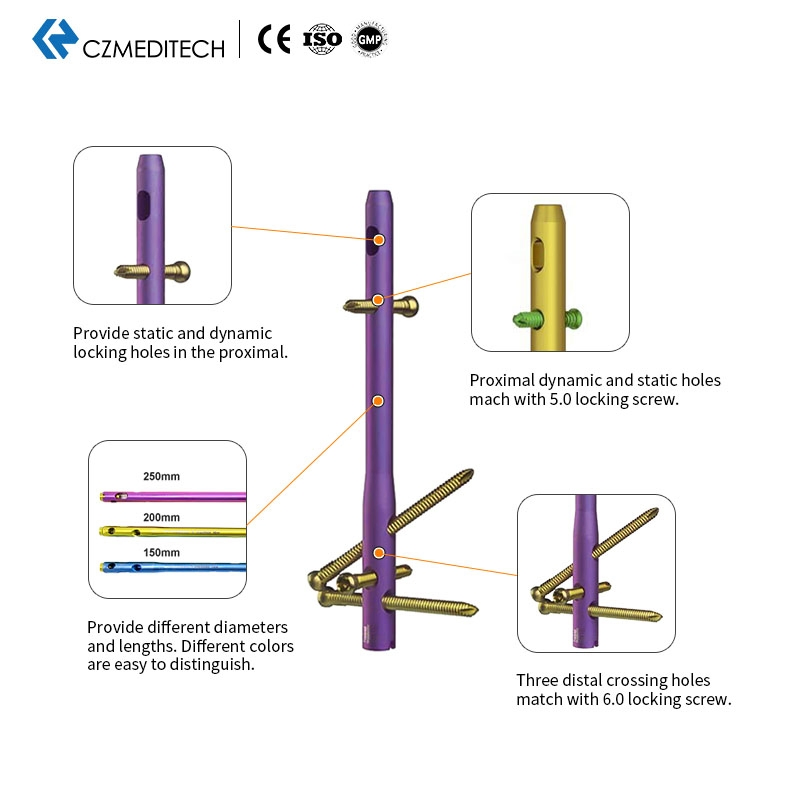
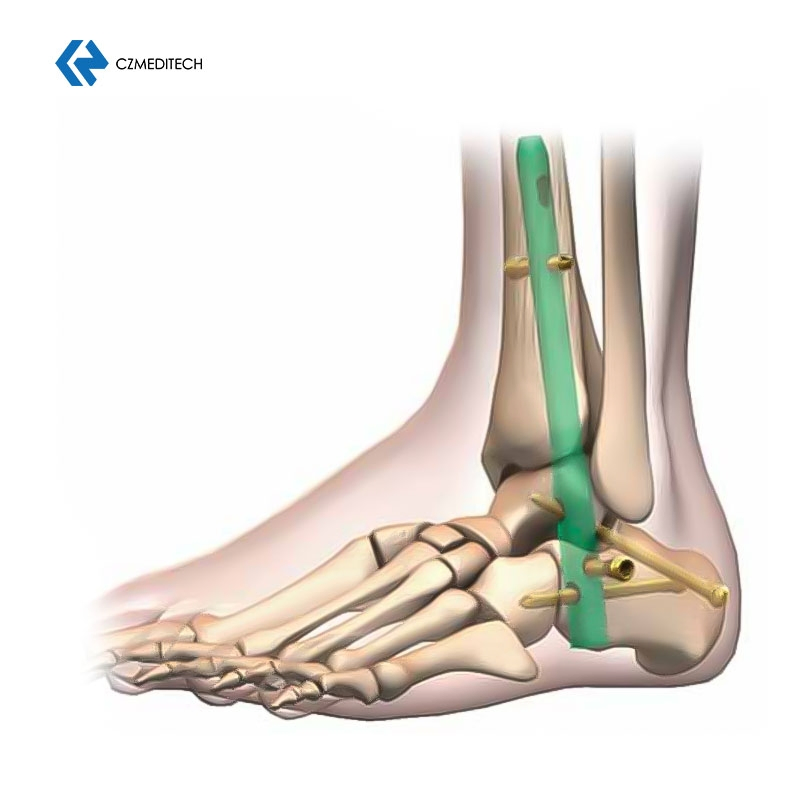
This titanium intramedullary nail is specifically designed for hindfoot and ankle fusion procedures, including tibiotalocalcaneal (TTC) arthrodesis. The interlocking design provides excellent rotational and axial stability, promoting successful fusion. Made from medical-grade titanium alloy (Ti6Al4V), it offers superior biocompatibility, strength, and MRI compatibility. Available in multiple sizes to accommodate patient anatomy.
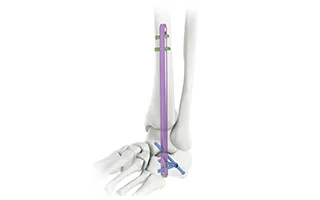






| Product Description | Available Sizes (Length) |
|---|---|
| Hindfoot Fusion Ankle Nail | 10mm Diameter: 150/200/225/250 mm (Left/Right) |
| 11mm Diameter: 150/200/225/250 mm (Left/Right) | |
| 12mm Diameter: 150/200/250 mm (Left/Right) | |
| 5.0mm Locking Screws | Lengths: 28mm to 60mm |
| 6.0mm Locking Screws | Lengths: 26mm to 100mm |
One-stop Procurement Solutions
As a leader in orthopedic implants and instruments manufacturing, we have successfully supplied to 2,500+ clients in 70+ countries for over 15 years. Our comprehensive product portfolio spans orthopedics, trauma care, veterinary medicine, and sterilization.

Professional Service: We provide a 1-year warranty. During this period, we offer repair or replacement services. All technical inquiries receive responses within 2 business days. Our systems are ISO 13485 and CE certified, ensuring international quality standards.