


1 / 4
 MGMT Orthopedic Implants
MGMT Orthopedic Implants








| Still deciding? Get samples of $ ! US$ 102/Piece Request Sample |

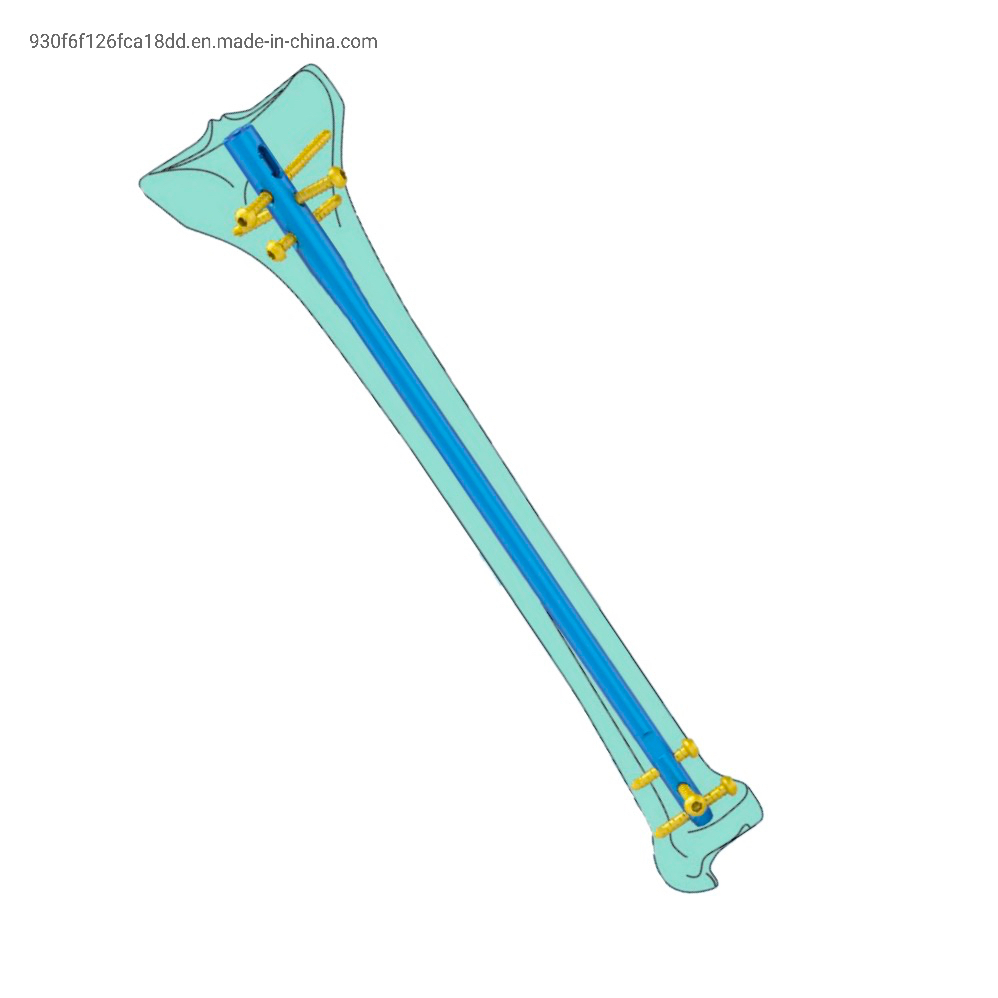
The Tibial Nail Advanced implants are intended for treatment of fractures in adults and adolescents (12-21) in which the growth plates have fused. Specifically, the implants are indicated for:

| Product name | Intramedullary Nail System Expert Tibial Interlocking Nail Suprapatellar Approach |
| Material | Titanium |
| Color | Grey , Blue , Pink , Red , Gold |
| Code | 6201 |
| Thickness | 8/9/10 mm |
| Length | 180-360 mm |
| Related Screw | 4.8mm lag screw , 4.35mm cortex screw |
| Package | Non Sterilized |