



 MGMT Orthopedic Implants
MGMT Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 500/Set Request Sample |

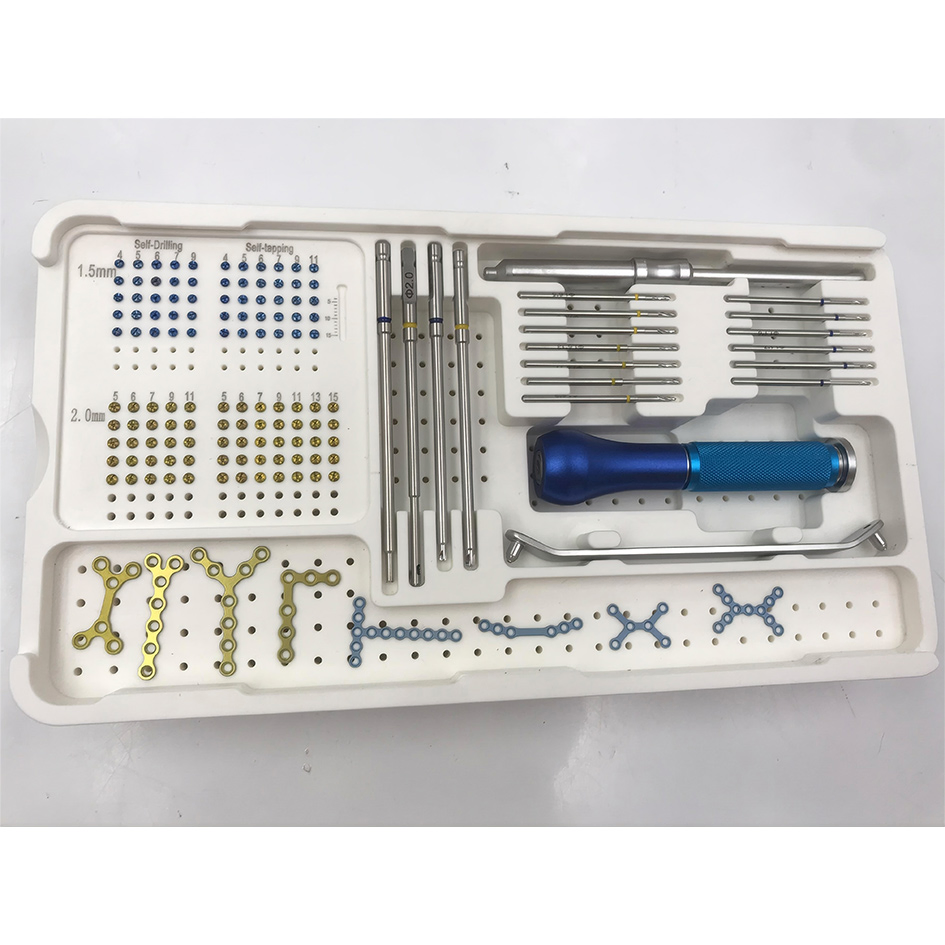
| System Name | No. | Code | Description | Qty |
|---|---|---|---|---|
| Instrument Set for 1.5/2.0mm System | 1 | 3128-0101 | Hollow Reamer 1.5mm | 1 |
| 2 | 3128-0102 | Hollow Reamer 2.0mm | 1 | |
| 3 | 3128-0103 | Screwdriver for 1.5mm Screw | 1 | |
| 4 | 3128-0104 | Screwdriver for 2.0mm Screw | 1 | |
| 5 | 3128-0105 | Depth Gauge 0-30mm | 1 | |
| 6 | 3128-0106~12 | Limited Drill Bit 1.6mm (5mm-15mm) | 7 | |
| 7 | 3128-0113~18 | Limited Drill Bit 1.1mm (4mm-11mm) | 6 | |
| 8 | 3128-0119 | Quick Coupling Handle | 1 | |
| 9 | 3128-0120 | Guider 1.1mm & 1.6mm | 1 | |
| 10 | 3128-0121 | Aluminium Box | 1 |

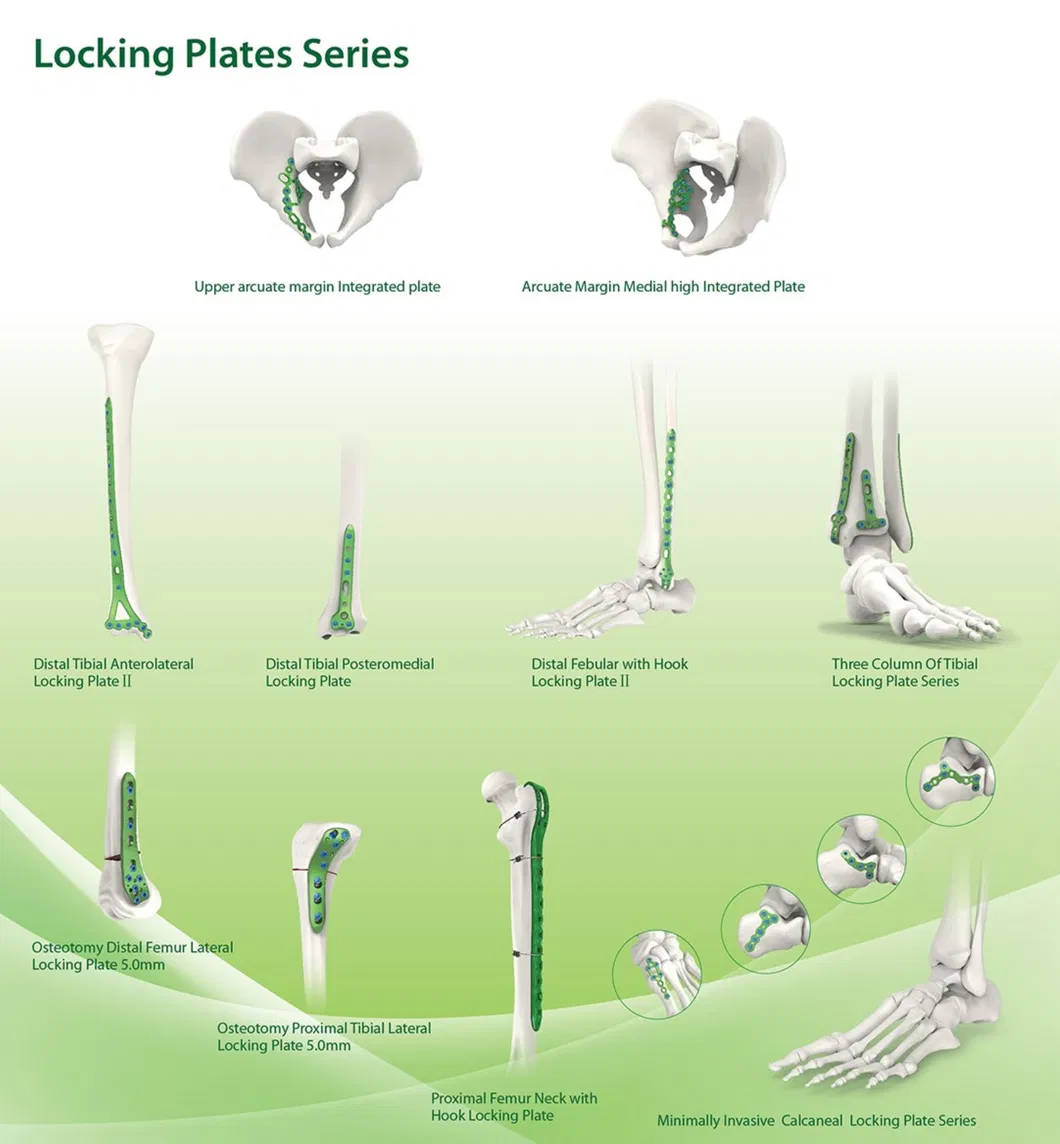






Established in 1958, the manufacturer is engaged in the development, manufacture, and sales of orthopedic implants and instruments. As a key enterprise for national medical apparatus and a director company of the Medical Apparatus Industry Association, the focus remains on orthopaedics implants, including spine pedicle screws, interlocking nails, locking plates, trauma plates, cannulated screws, external fixators, and hip prosthesis.
Equipped with imported CNC machines and high-precision digital control systems, the production facility includes a 100,000 Grade axenic purification chamber to ensure high-quality standards. The company was among the first to pass ISO 9001 quality system certification, with products receiving national silver awards and invention prizes.
