




 MGMT Orthopedic Implants
MGMT Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 88/Piece Request Sample |
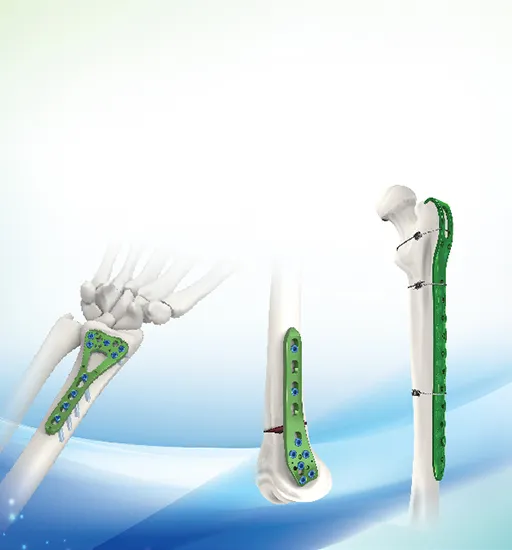
| Multi-axial Distal Femur Lateral Locking Plate 341 | ||
|---|---|---|
| P/N | Material | Holes |
| 34104-34113 L/R | Titanium | 4,5,6,7,8,9,10,11,12,13 holes |
| Use 5.0 locking screw, 5.0 cortical self tapping screw | ||









Established in 1958, our enterprise is dedicated to the development, manufacture, and sales of orthopedic implants and instruments. As a key player in the national medical apparatus sector and a director company of the Medical Apparatus Industry Association, we offer a comprehensive product line including spine pedical screws, interlocking nails, locking plates/screws, trauma plates, cannulated screws, external fixators, hip prosthesis, and surgical instruments.
Our products are characterized by multi-varieties and complete specifications. All medical apparatus have passed rigorous approval and authentication by national and provincial superintendent offices. The production facility features advanced equipment including imported CNC, high-precision digital control machines, and a 100,000 Grade axenic purification chamber to ensure superior product quality.
