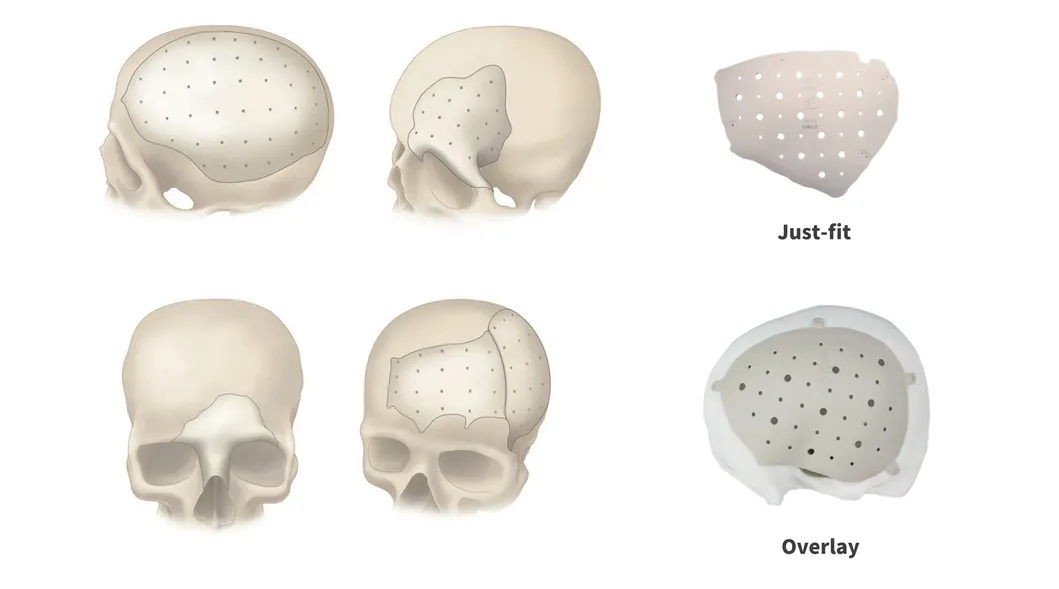
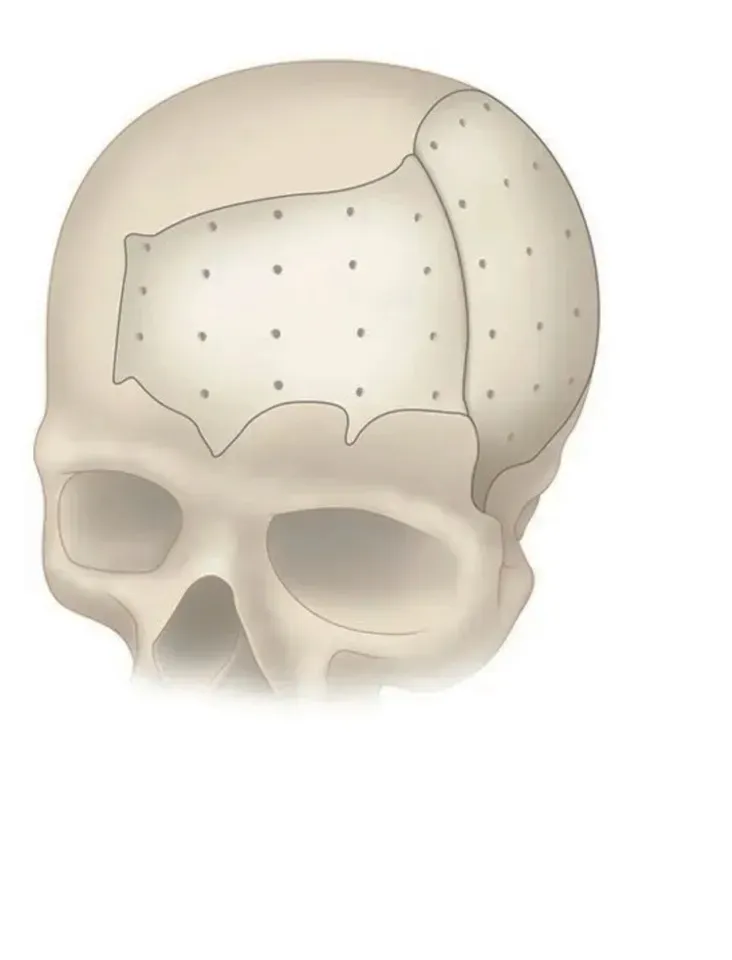
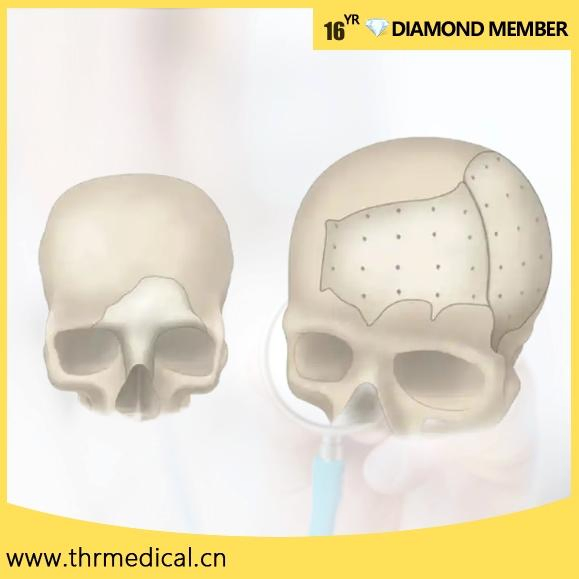
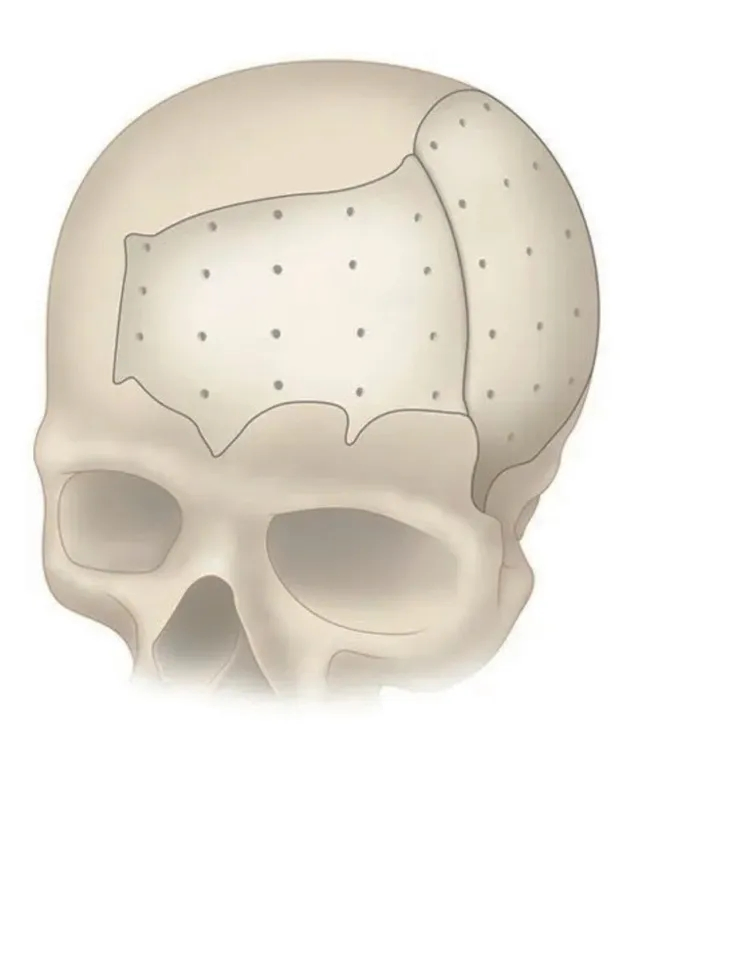
PEEK Patient Specific Implant (PSI)
PEEK (Polyether Ether Ketone) Patient Specific Implant provides an improved solution for patients with a cranio-maxillofacial defect. Our technology and craftsmanship produce a perfect match to the specific anatomical requirement in a custom implant.
PEEK can be designed and provided in single or multiple pieces with just-fit or over-lay concept as indicated above. Additionally, this patient specific implant can be reliably fixated using cranial fixation systems.
Excellent Biocompatibility
Non-cytotoxic, corrosion resistance, non-carcinogenic, non-conductive, and hydrolysis resistance. Good biological characteristics and histocompatibility. Capable to sustain repeated steam, ETO sterilization, and Gamma irradiation.
Outstanding Bio-Mechanical Character
Exceeding tensile strength to the native bone and provide better protection to the cerebrum. Comparable mechanical strength to the artificial bone and capable to distract the stress effectively when deformity occurs.
Better Imaging Compatibility
Non-Magnetic and highly translucent to X-ray. CT/MRI imaging diagnosis without scattering or artifacts.
Perfect Contour Reconstruction
Precisely match the natural contour and convenient to implant. CAD/CAM technology allows to restore the defect models and reconstruct the natural contour of the brain.
Frequently Asked Questions
What is the primary material used for these implants?
The implants are made from PEEK (Polyether Ether Ketone) and medical-grade titanium, known for their excellent biocompatibility and mechanical strength.
Are PEEK implants compatible with MRI and CT scans?
Yes, PEEK is non-magnetic and highly translucent to X-rays, allowing for clear CT and MRI imaging without the artifacts or scattering often caused by other materials.
How is a "perfect fit" achieved for each patient?
We utilize advanced CAD/CAM technology to restore defect models based on patient-specific data, ensuring the implant precisely matches the natural contour of the skull or face.
Can the implants be sterilized?
Yes, the materials are designed to sustain repeated steam sterilization, ETO sterilization, and Gamma irradiation without degrading.
What are the clinical advantages of PEEK over other materials?
PEEK offers mechanical strength comparable to native bone, superior imaging compatibility, and a lower thermal conductivity, which provides better protection to the brain.
Is OEM service available for these products?
Yes, OEM services are accepted to meet specific clinical or institutional requirements.



 MGMT Orthopedic Implants
MGMT Orthopedic Implants