 MGMT Orthopedic Implants
MGMT Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 40/Piece Request Sample |

| Product | Length (mm) | Material |
|---|---|---|
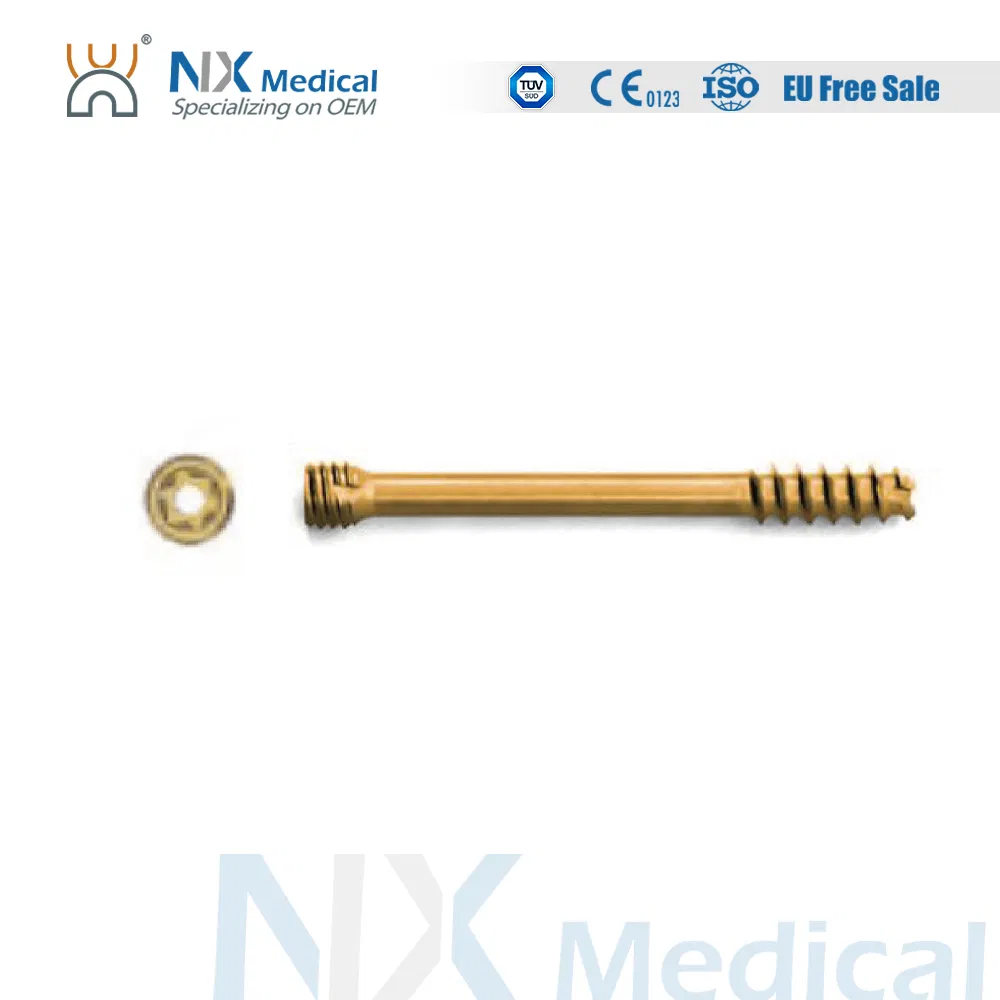
| φ2.5mm Headless Compression Screw - I | 12/14/16/18/20/22/24/26/28/30 | Titanium Alloy |
| φ3.0mm Headless Compression Screw - I | 14/16/18/20/22/24/26/28/30/32/34/36/38/40 | Titanium Alloy |
| φ3.5mm Headless Compression Screw - I | 16 to 60 (2mm increments) | Titanium Alloy |
| φ4.0mm Headless Compression Screw - I | 20 to 60 (2mm increments) | Titanium Alloy |
| φ4.5mm Headless Compression Screw - I | 30 to 70 (various) | Titanium Alloy |
| φ6.5mm Headless Compression Screw - I | 30 to 100 (5mm increments) | Titanium Alloy |


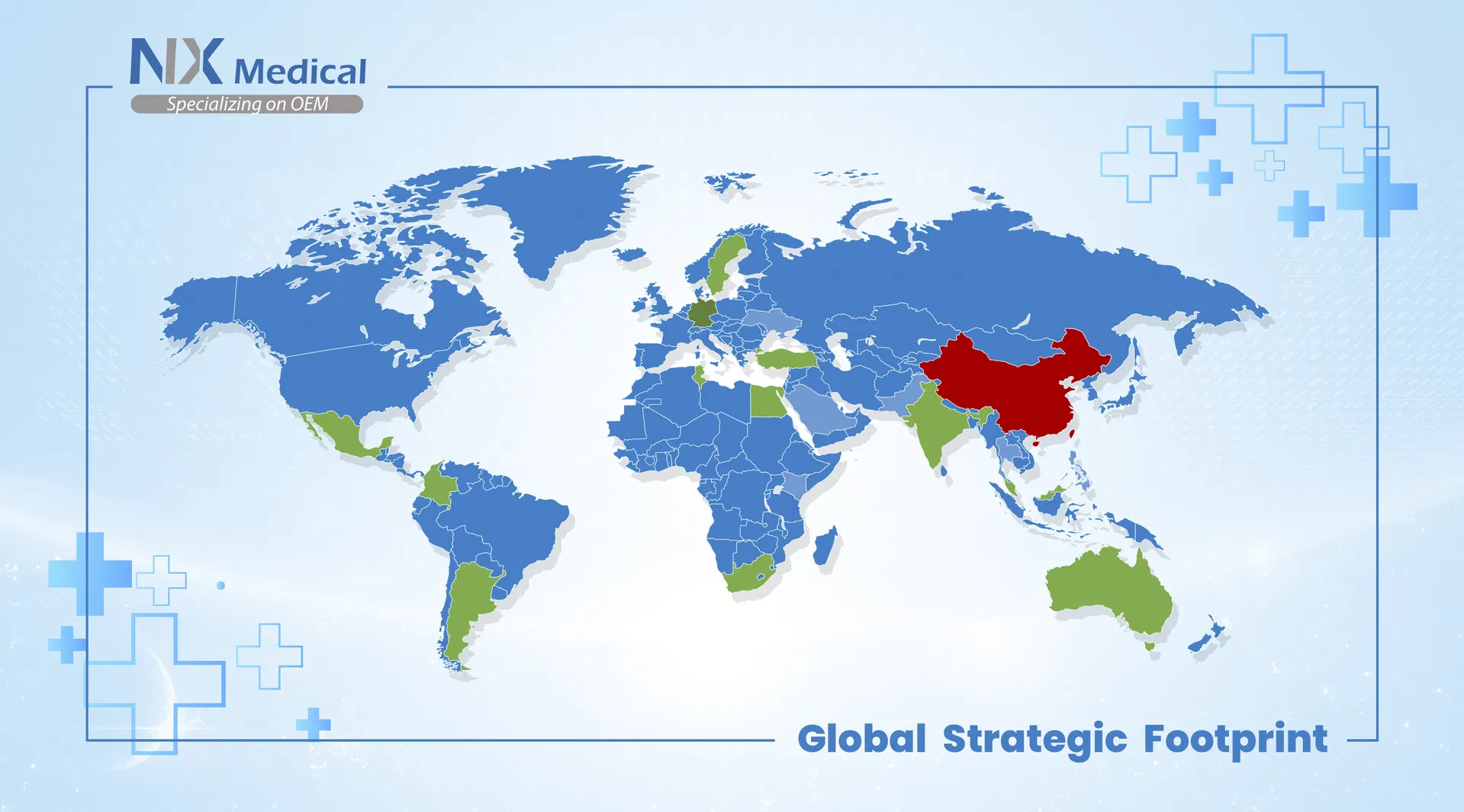
Our facility is one of the leading Orthopedic product manufacturers located in the East of China. We provide a comprehensive range of cost-effective surgical and reliable therapeutic solutions to Orthopedic surgeons and patients worldwide. Our offerings include superior trauma implants, spinal implants, and surgical instruments.
Trauma System: Locking plates, Intramedullary nails, cannulated headless screws, and cable systems.
Spinal System: Cervical plates, pedicle screws, PEEK cages, and expandable cages.
We maintain a strict internal quality system to fulfill frequent self-audits. Our products comply with ISO13485, and are certified by TUV CE0123 and NMPA, ensuring the safest and most advanced technologies for rapid recovery and pain-free life after surgery.