



1 / 5
 MGMT Orthopedic Implants
MGMT Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 80/Piece Request Sample |


Key Features and Benefits:
| Product | Size (mm) | Direction | Material |
|---|---|---|---|
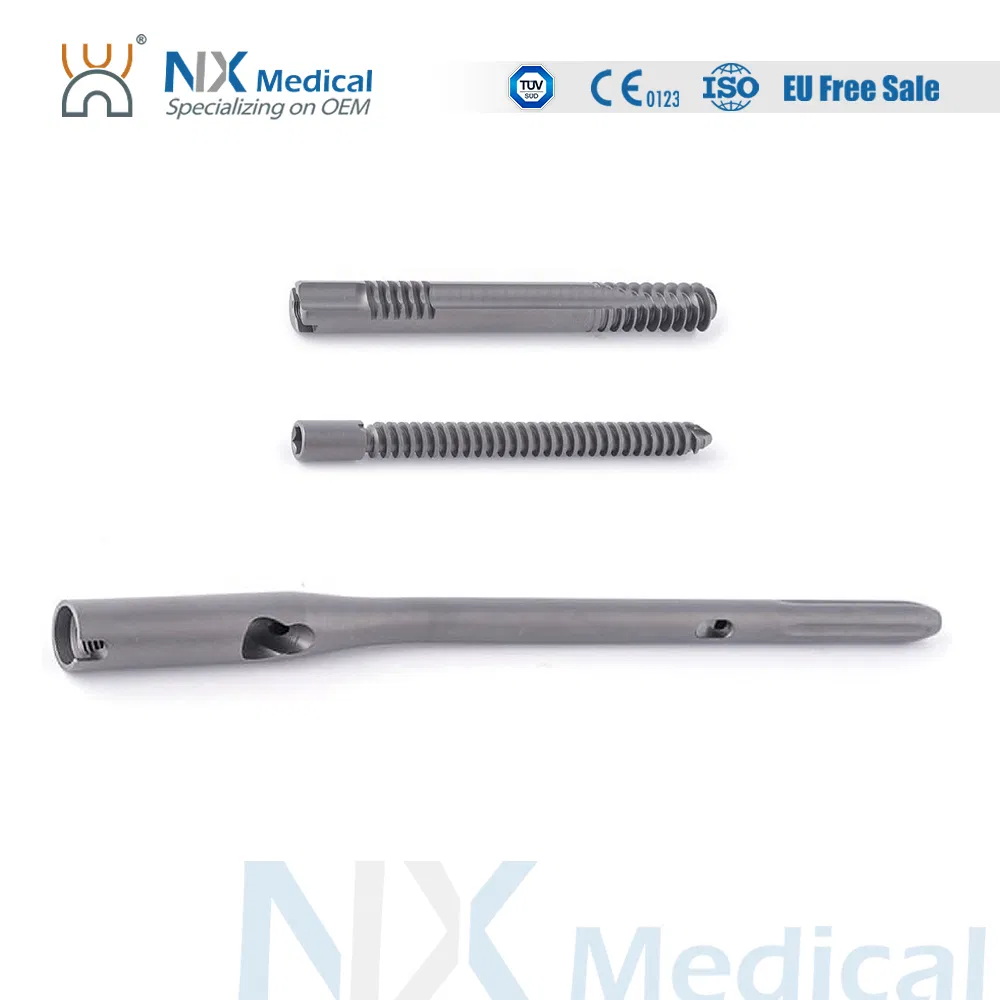
| PFIA Nail | 9 x 180 / 200 / 220 / 240 | Standard | Titanium Alloy (TA) |
| 9 x 320 / 340 / 360 / 380 / 400 / 420 / 440 | Left / Right | Titanium Alloy (TA) | |
| 10 x 180 / 200 / 220 / 240 | Standard | Titanium Alloy (TA) | |
| 11 x 320 / 340 / 360 / 380 / 400 / 420 / 440 | Left / Right | Titanium Alloy (TA) | |
| 13 x 320 / 340 / 360 / 380 / 400 / 420 / 440 | Left / Right | Titanium Alloy (TA) | |
| Lag Screw | φ10.6mm (Length: 70 - 120mm) | - | TA |
| Sub Lag Screw | φ7.0mm (Length: 65 - 115mm) | - | TA |
| End Cap | Extension: 0 / 5 / 10 / 15mm | - | TA |
