


1 / 4
 MGMT Orthopedic Implants
MGMT Orthopedic Implants








| Still deciding? Get samples of $ ! US$ 40/Piece Request Sample |


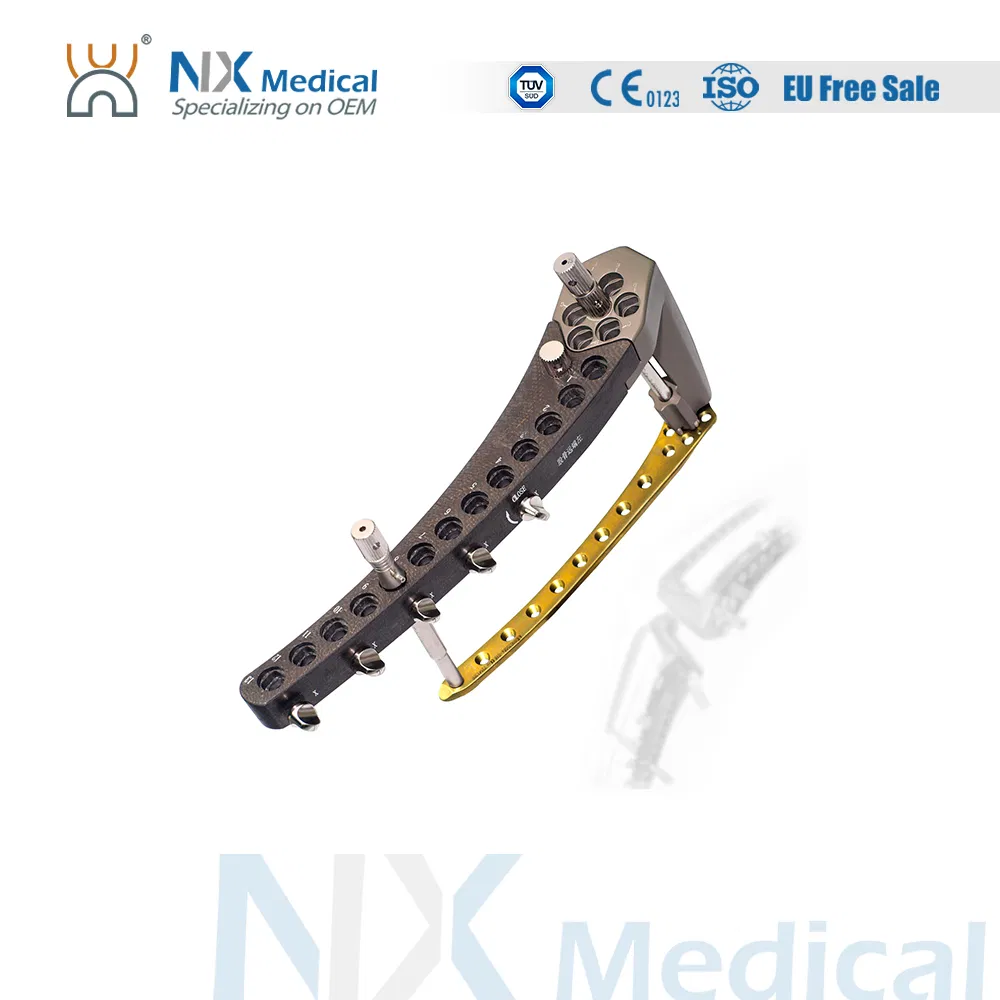
| Product Name | Holes | Direction | Material | Length (mm) |
|---|---|---|---|---|
| 5.0mm Distal Femoral Liss LCP | 7+5H | Left/Right | PT | 158 |
| 7+9H | Left/Right | PT | 238 | |
| 7+13H | Left/Right | PT | 318 | |
| 7+17H | Left/Right | PT | 398 | |
| 5.0mm Proximal Lateral Tibial Liss LCP | 4+5H | Left/Right | PT | 138 / 98 |
| 4+13H | Left/Right | PT | 298 / 258 |

Our facility is a leading orthopedic product manufacturer located in East China, providing comprehensive, cost-effective surgical and reliable therapeutic solutions to surgeons and patients worldwide. We offer superior orthopedic trauma implants, spinal implants, and high-precision surgical instruments.
Commitment to quality is our core value. We have established a strict internal quality system to fulfill frequent self-audits. Externally, we comply with ISO13485, and all orthopedic products are certified by TUV CE0123 and NMPA.