Manufacturing & Quality Control
As a national high-tech enterprise, R&D investment exceeds 8% of total sales annually. We specialize in transforming clinical experience into innovative medical solutions with numerous national patents. Our key technologies include revolutionary drilling systems, sternal fixation, and rib plates, widely recognized as industry benchmarks.
Operations strictly follow the ISO13485 quality system and CE MDD 93/42/EEC directives. Our facility features German-imported and American-imported machining centers, and Japanese automatic lathes. The Testing Center includes a biomechanical laboratory and physical-chemical testing center to ensure raw material control and product performance.
Certifications & Shipping
Products are exported globally to countries including Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. We also serve as a major OEM supplier for the largest global players in the orthopaedic industry.
Frequently Asked Questions
Is customized design available for orthopaedic implants?
Yes, we have a professional team to handle customized items based on provided drawings or samples.
What certifications do your spinal fixation systems hold?
Our products are certified with CE, ISO, and FDA for the Spinal System, ensuring international quality standards.
Can I request samples for quality and market testing?
Yes, we welcome sample orders to test our product quality and service before placing bulk orders.
What is the typical lead time for orders?
Sample orders are typically processed within 7 days, while standard bulk orders are delivered within 25 days.
How do you ensure the quality of raw materials?
We operate a specialized Testing Center with physical-chemical laboratories to strictly control raw material procurement and product performance analysis.
Do you support international shipping and import guidance?
Yes, we assist and guide clients through the shipment and import process, especially for those new to international business.

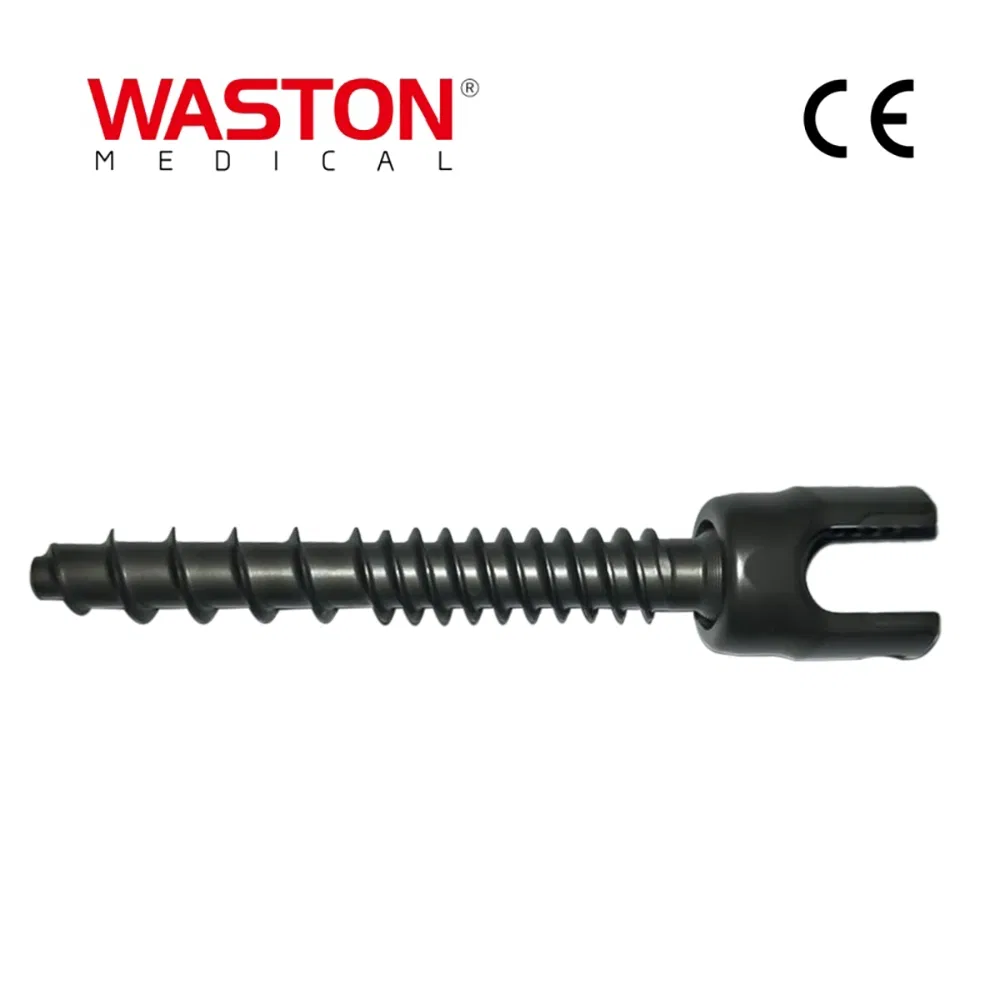
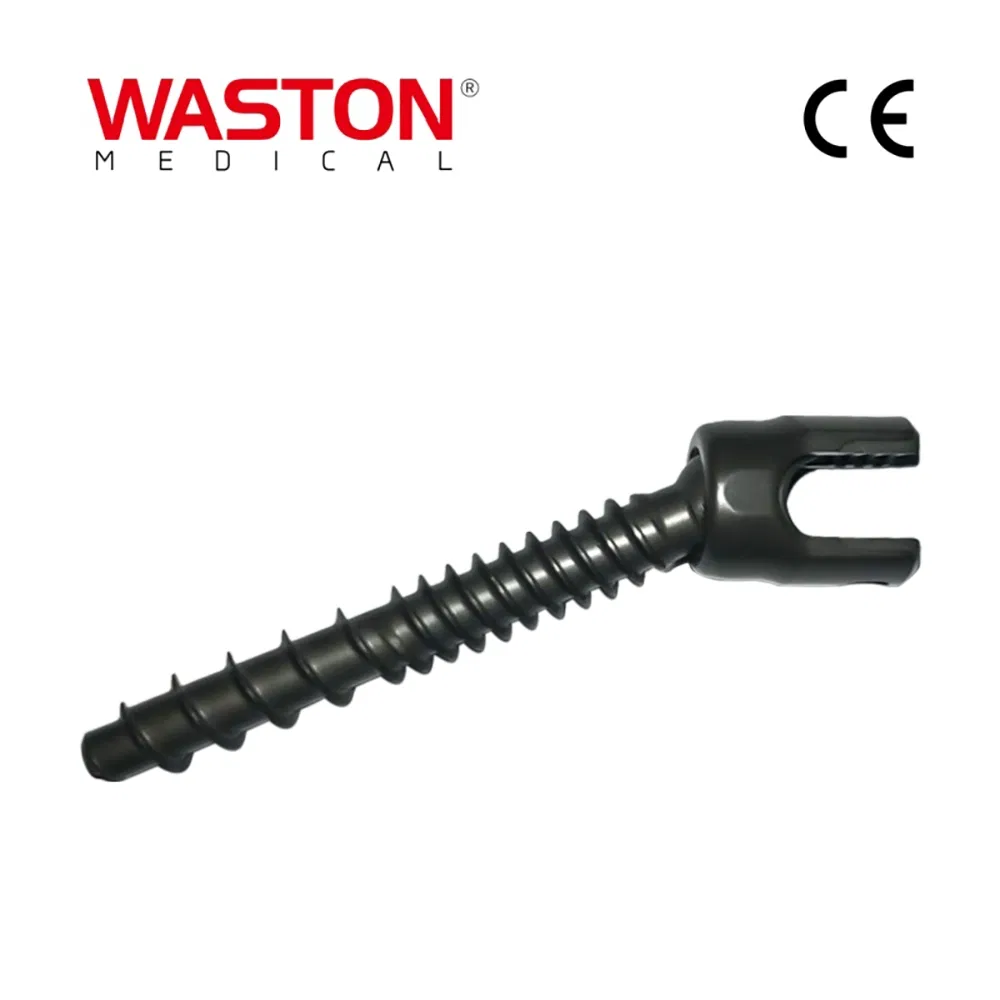
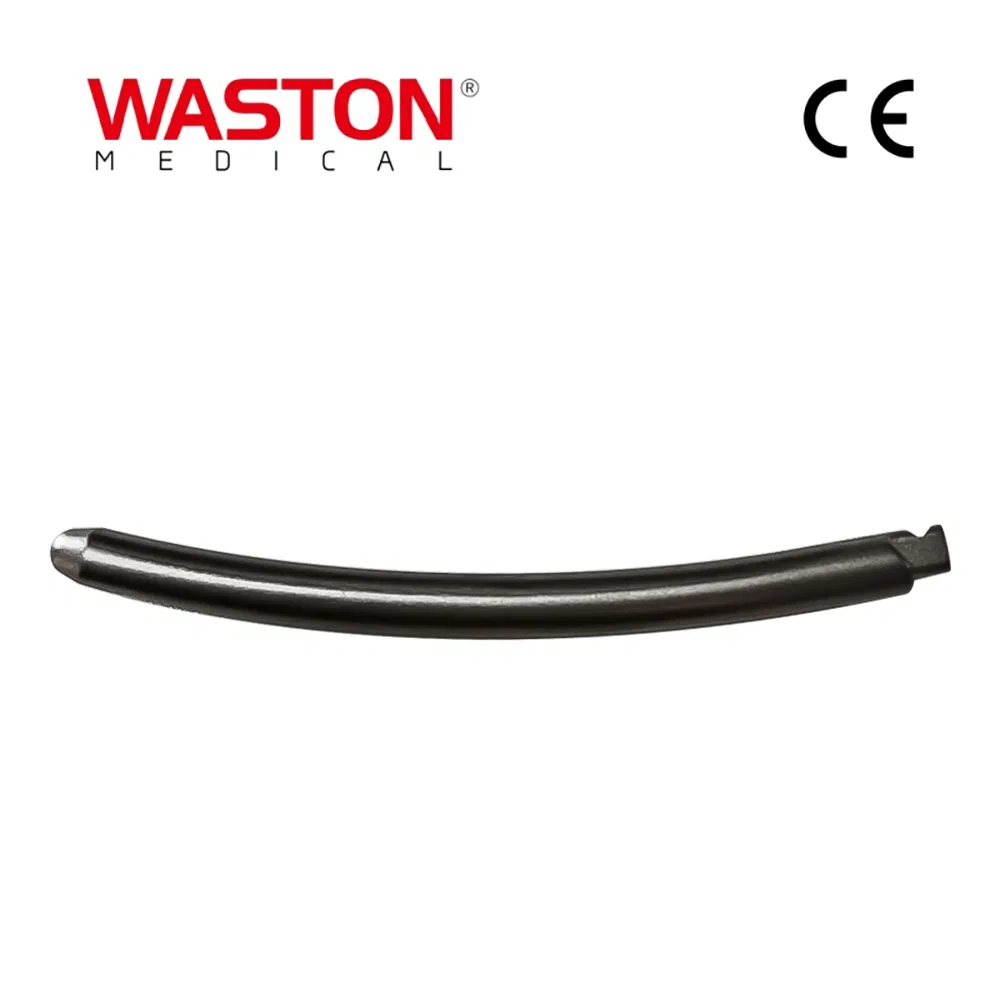

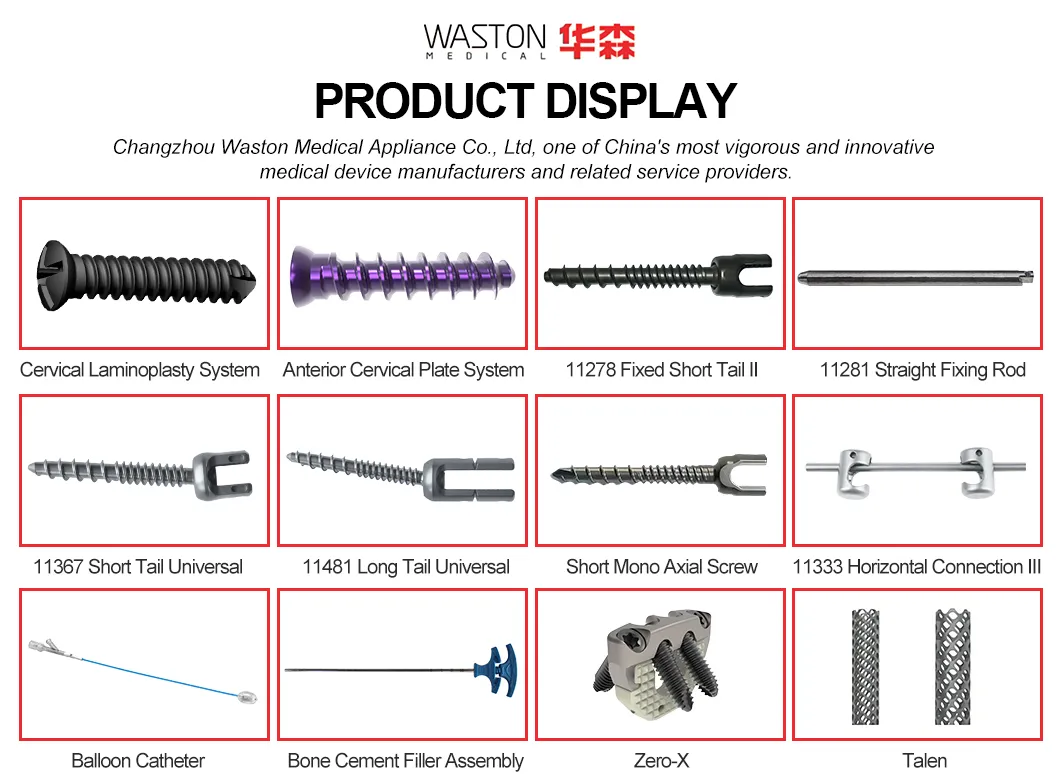
 MGMT Orthopedic Implants
MGMT Orthopedic Implants