




1 / 5
 MGMT Orthopedic Implants
MGMT Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 100/Piece Request Sample |


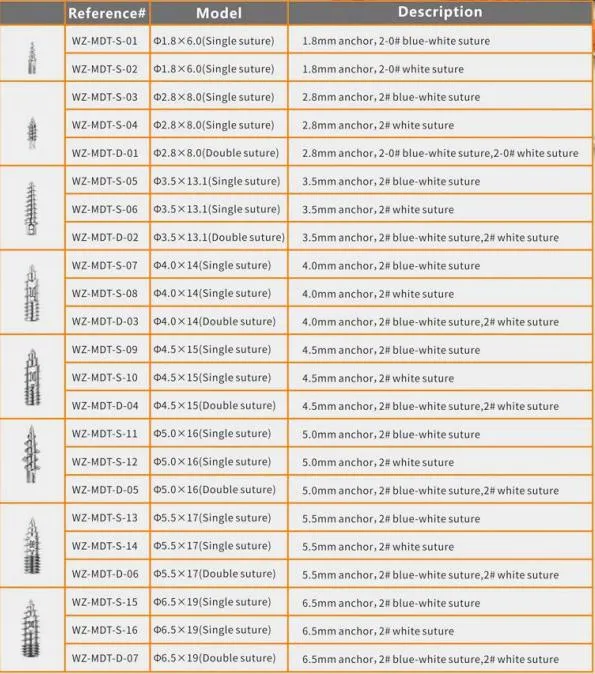
| Product Name | Non-bioabsorbable Suture Anchor |
| Ctn Size | 42*22.5*25.5CM |
| G.W./CTN | 1.5KG-1.8KG |
| Certificates | CE & ISO13485 |
| Period of Validity | 3 Years |
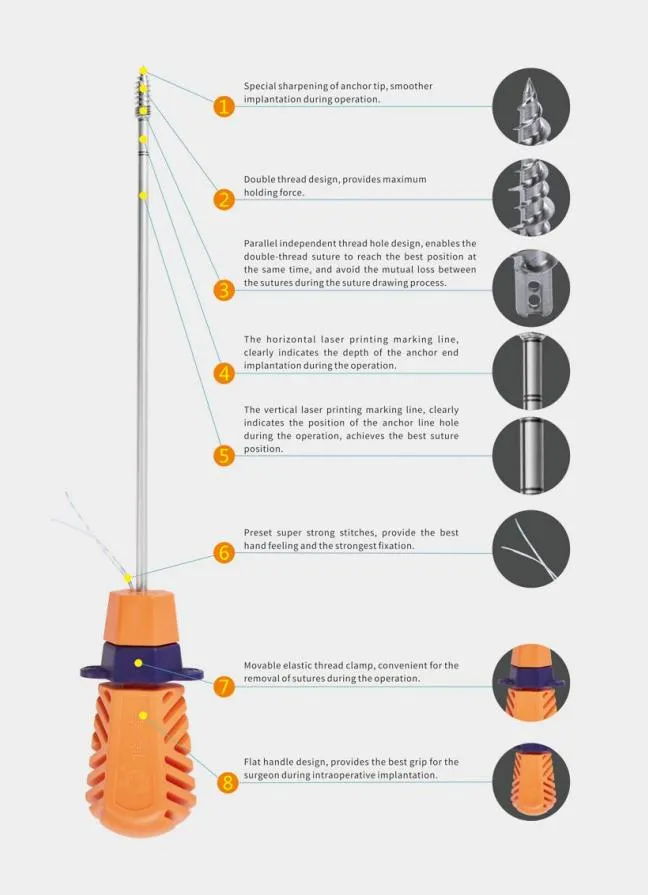
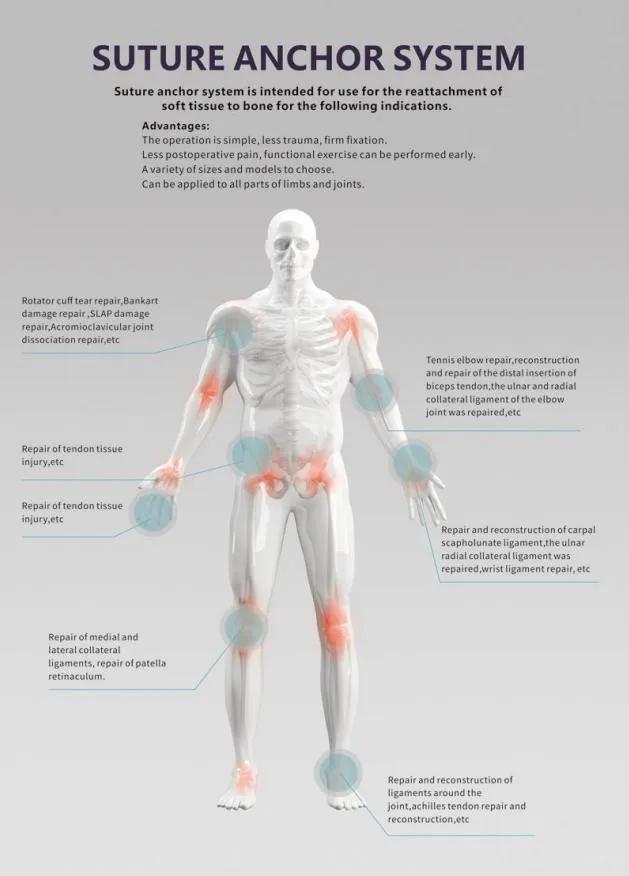


We are a leading manufacturer in China specializing in Disposable Surgical Lavage Systems for effective debridement following orthopedic procedures of trauma and joint arthroplasty. All our products are certified by CE & ISO13485.
Our Surgical Lavage System combines convenience and power for the removal of contaminated water, necrotic tissue, bone debris, wound pollutants, and foreign material. It provides controlled pressure for efficient cleansing and irrigation on flat wound surfaces, osteotomy surfaces, or the intra-medullary space.
Products are continuously improved based on clinical research and surgeon feedback. Noise and weight (currently only 772g) are minimized, making them highly favorable for modern orthopedic procedures.