



1 / 5
 MGMT Orthopedic Implants
MGMT Orthopedic Implants










| Customization: | Available |
|---|---|
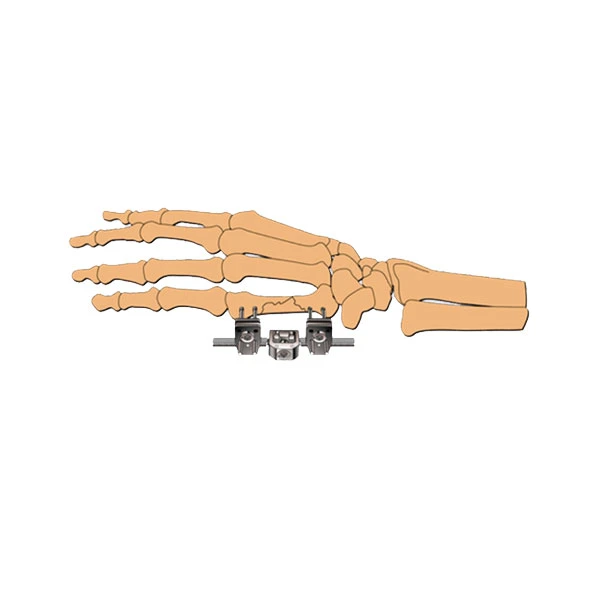
| Type: | External Fixation |
| Application: | Orthopedic |
Application: Used for patients with fractures of limbs.
Suitable Situations:
| Code | Specification | Note |
| D501 | Long 85mm | - |
| D502 | Middle 75mm | - |
| D503 | Short 65mm | - |
| Schanz Pin | ||
| Diameter(mm) 2.0 | Specification(mm) screw length/thread length | |
Product Details:




