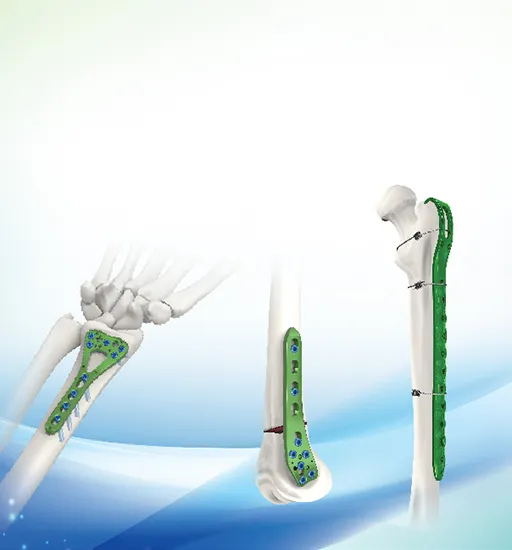
📋 Overview & Basic Information
Model NO.311
MaterialTitanium
FeatureDisposable
CertificationCE, ISO13485
GroupAdult
UsageBone Fracture Fixation
ColorGrey
Surface(Ti)Micro-Arc Oxidation
OEM/ODMAccept
Specification3/4/5/6/7/8/9/10 holes
🏭 Professional Manufacturing & Exhibition
Established in 1958, our enterprise is dedicated to the development, manufacture, and sales of orthopedic implants and instruments. We are recognized as a key enterprise for medical apparatus, providing a complete range of spine pedicle screws, interlocking nails, locking plates, trauma screws, and surgical instruments.
Equipped with advanced CNC machines and a 100,000 Grade sterile purification chamber, we ensure high-quality standards. Our products have earned numerous technology awards and are trusted globally for trauma fixation.
❓ Frequently Asked Questions
1. What types of orthopedic implants do you manufacture?
We specialize in metal bone plates and screw systems, metallic cannulated bore screws, interlocking intramedullary nails, spinal fixation devices, and external fixation systems.
2. How is the quality of the implants guaranteed?
Our products are manufactured according to EU CE standards. We conduct pre-production sample testing and a final inspection of every batch before shipment to ensure clinical safety.
3. What materials are used in your locking plates?
Most of our trauma plates and screws are made from high-grade Pure Titanium or Titanium Alloy, featuring Micro-Arc Oxidation surfaces for better biocompatibility.
4. Why should we choose your products over other suppliers?
With over 50 years of experience in R&D and manufacturing, we offer a strict quality control system, full CE/ISO/GMP certifications, and comprehensive technical support.
5. What delivery and payment terms are accepted?
We accept various delivery terms including FOB, CIF, EXW, and Express Delivery. Payments can be made via T/T, L/C, Western Union, or Escrow in USD, EUR, or CNY.
6. Do you offer OEM/ODM services for specific clinical needs?
Yes, we accept OEM and ODM requests to customize specifications, hole counts, and designs based on specific surgical requirements.





 MGMT Orthopedic Implants
MGMT Orthopedic Implants