


 MGMT Orthopedic Implants
MGMT Orthopedic Implants






| Still deciding? Get samples of $ ! US$ 85/Piece Request Sample |
The Interface Screw is designed for use in ACL reconstruction surgeries to secure tendon or ligament grafts in the femoral and tibial bone tunnels. It provides a strong and reliable fixation by compressing the tendon or ligament graft directly against the bone tunnel wall.
The screw's unique mechanism ensures optimal stability and a firm attachment between the soft tissue graft and the bone. The system includes a conical screw for easy insertion and a protective sheath that shields the soft tissue and reduces the risk of displacement. Made from PEEK (Polyetheretherketone), it mimics the elasticity of natural bone for superior healing performance.
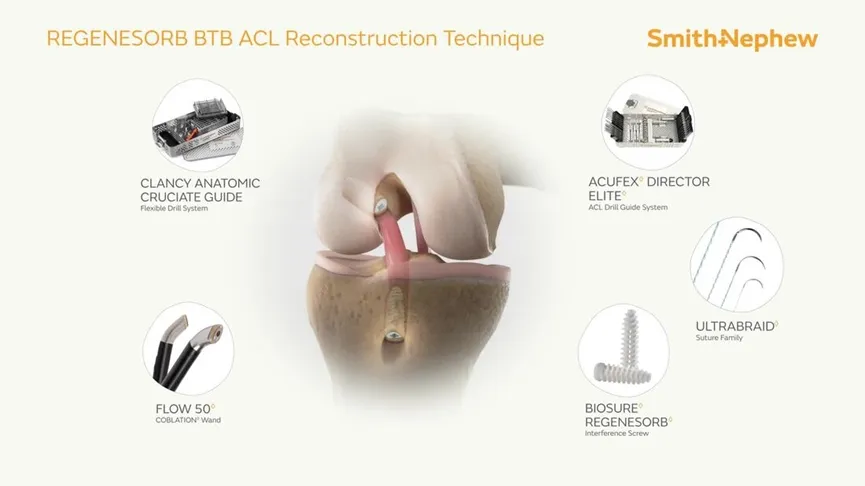


This Interface Screw System is primarily used for ACL reconstruction surgeries. It provides secure fixation for soft tissue grafts or bone-tendon grafts in the knee joint, ensuring effective healing and post-operative recovery.