1 / 5
 MGMT Orthopedic Implants
MGMT Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |
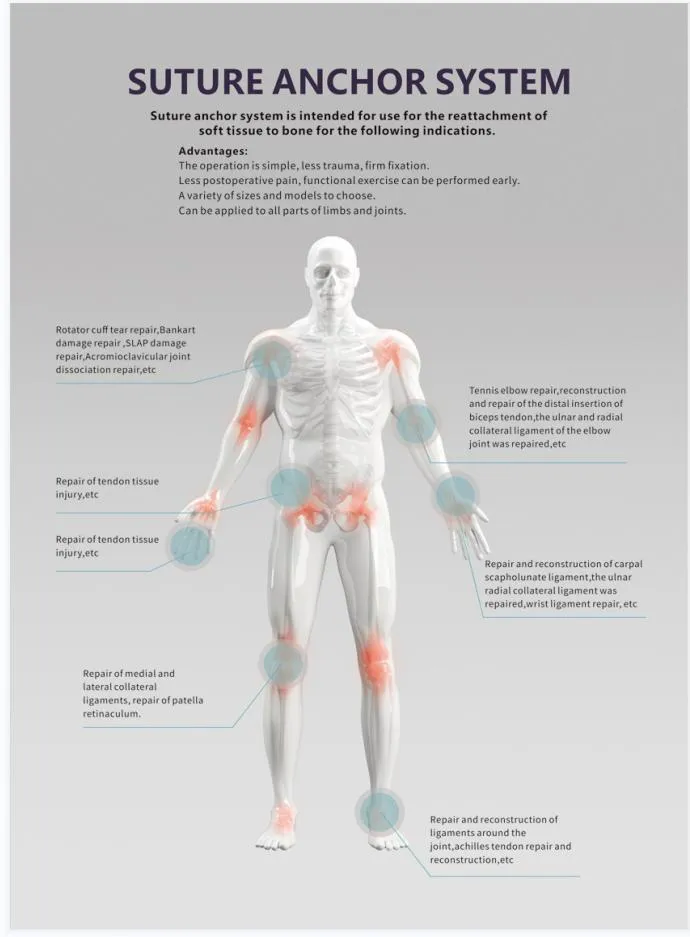
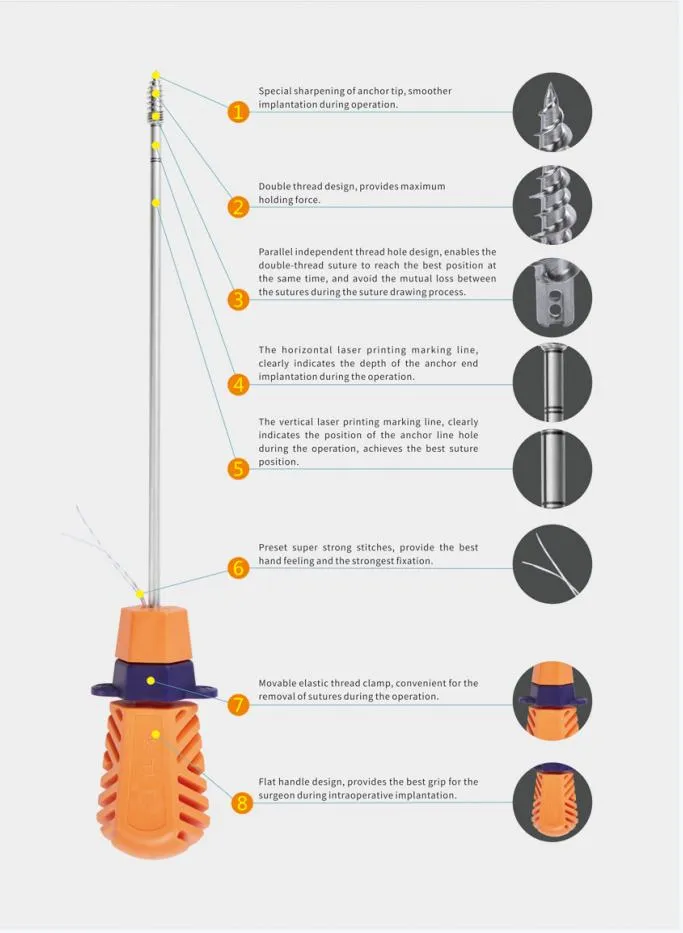
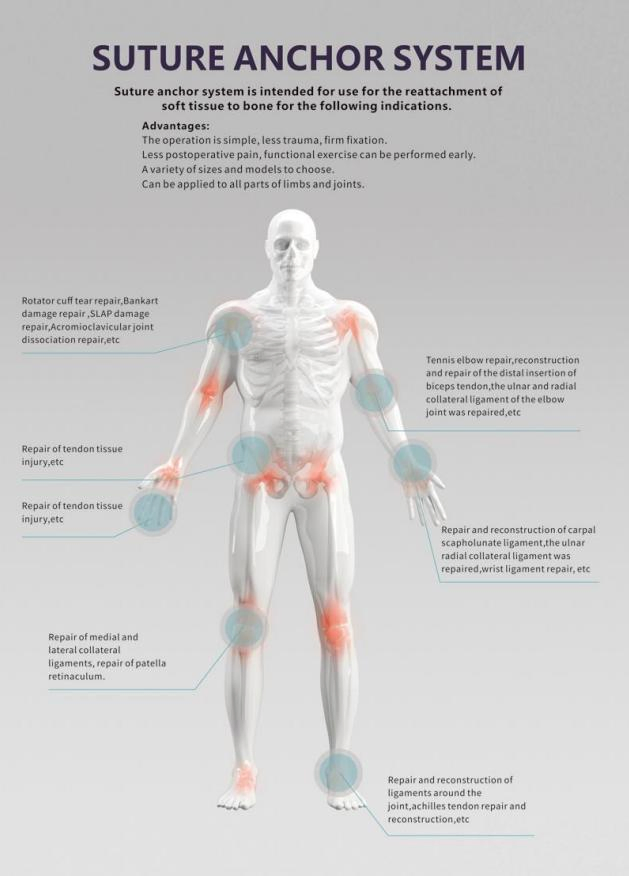
The Suture Anchor System is a high-precision medical device consisting of an anchor, nonabsorbable suture, and an inserter. It is designed specifically for sports medicine and orthopedic procedures to facilitate the repair of ligaments and tendons.





| Item Property | Details |
|---|---|
| Product Name | Suture Anchor System |
| Material | Titanium Alloy (Ti6Al4V) |
| Specification | Diameter 1.8 x 6mm |
| Classification | Class II Medical Instrument |
| Packaging | Sterile Blister Box (1pc/package) |
| Application | Orthopedic Surgery / Implantology |
| Properties | Implant Materials & Artificial Organs |