1 / 5
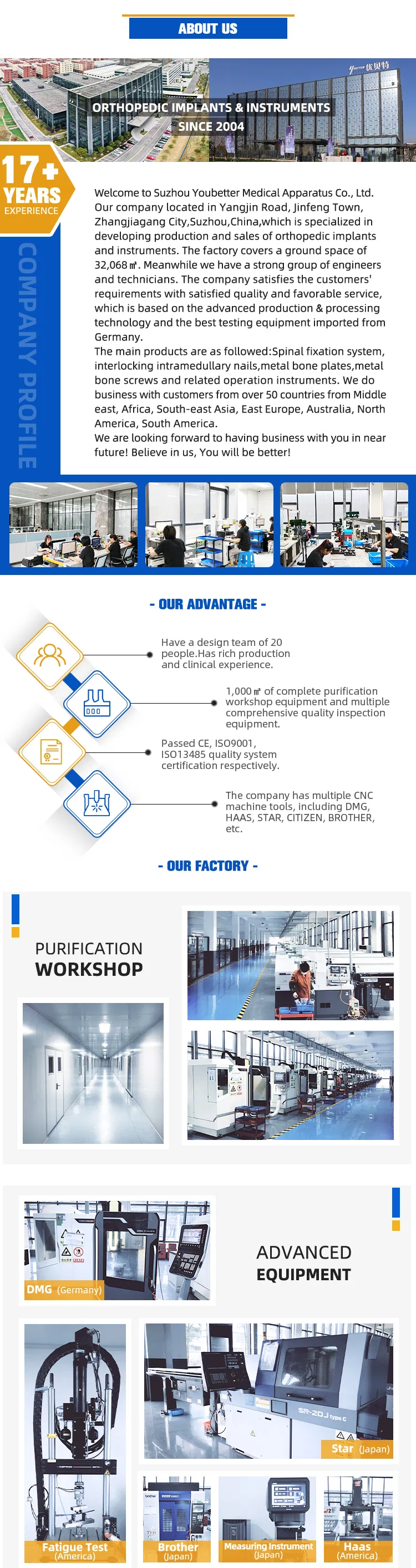
 MGMT Orthopedic Implants
MGMT Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 15/Piece Request Sample |
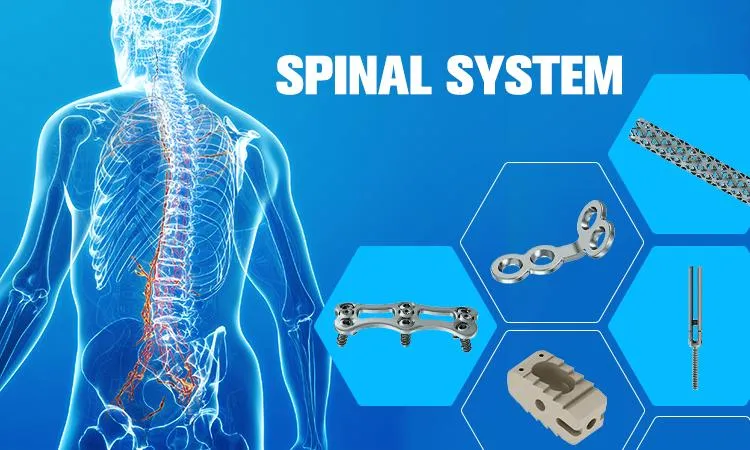

Advanced 5.5/6.0 Screw-Rod System for Spinal Internal Fixation
This mature implant system offers a versatile choice between sterile and non-sterile packaging, tailored to the high standards of medical professionals. Featuring specially designed hooks, rods, and screws, this system has significantly advanced spinal surgery techniques, providing robust solutions for complex spinal conditions.
The pedicle screw is engineered with a variable pitch thread, ensuring stable and firm fixation, which is critical for successful clinical outcomes and patient recovery.


| Product Name | Spinal Internal Fixation 5.5/6.0 U-Multi-Axial Reduction Pedicle Screw |
| Material | High-Grade Titanium |
| Available Colors | Gold, Blue, Grey, Pink, etc. |
| Related Rod Diameter | 5.5mm / 6.0mm |
| Screw Diameter | 4.5 / 5.0 / 5.5 / 6.0 / 6.5 / 7.0 mm |
| Screw Length | 30 / 35 / 40 / 45 / 50 / 55 / 60 mm |
| Certificates | CE / ISO 13485 & 9001 |
| Packaging Options | Non-Sterilized / Sterilized |