
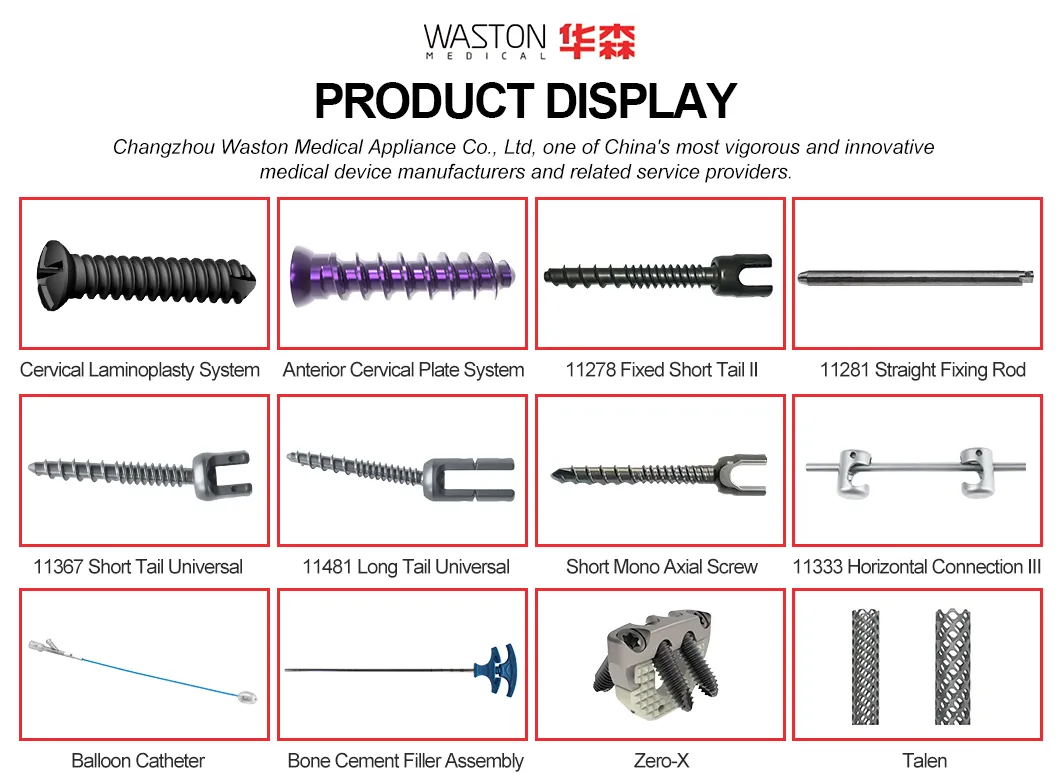
 MGMT Orthopedic Implants
MGMT Orthopedic Implants










| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
✔ Immediate lamina supporting in surgery, ensuring expanded canal plasty maintenance.
✔ Lower dentate supporting plate provides superior stable force.
✔ Arc openings designed for better anatomical inclusion.
✔ High plasticity for slight moulding during operative procedures.
★ Stable support for vertebral body decompression.
★ Four-hole design optimized for diverse surgical requirements.
★ Unique structural angle enhances vertebral body stability.

| PRODUCT NAME | PRODUCT CODE | DIA | DATA |
|---|---|---|---|
| NEULEN Laminoplasty Plate | 11101012 | / | H=12 |
| 11101014 | / | H=14 | |
| 11101016 | / | H=16 | |
| NEULEN Screw (Cross self-tapping) | 10511306 | φ2.5 | L=6 |
| 10511308 | φ2.5 | L=8 | |
| 10511310 | φ2.5 | L=10 |

As a national high-tech enterprise, the manufacturer invests over 8% of total sales in R&D annually. This commitment transforms clinical expertise into innovative medical solutions, holding numerous national invention and utility patents.


Operations are strictly governed by the ISO13485 quality system and comply with CE MDD 93/42/EEC directives. Production utilizes German, American, and Japanese high-precision machining centers to ensure world-class product quality.


